All published articles of this journal are available on ScienceDirect.
Translating Resveratrol Research: A Systematic Review on Patents and Clinical Trials (2000–2025)
Abstract
Introduction
Resveratrol (3,4′,5-trihydroxy-stilbene), a naturally occurring polyphenolic stilbenoid present in grapes, red wine, peanuts, and berries, has garnered immense scientific interest over the last two decades due to its multifaceted pharmacological properties. Its active trans-isomer (RSV) has demonstrated antioxidant, anti-inflammatory, anti-diabetic, cardioprotective, neuroprotective, anti-aging, anti-obesity, and anticancer effects by modulating many key molecular targets. Despite encouraging preclinical findings, the clinical translation of RSV has been hampered by poor systemic absorption, rapid metabolism, and low oral bioavailability.
Methods
This systematic review adheres to the PRISMA 2020 guidelines and provides a comprehensive analysis of data obtained from PubMed, Scopus, Web of Science, Google Scholar, Google Patents, and ClinicalTrials.gov, spanning the period from 2000 to 2025. The focus was on patents and clinical trials investigating RSV and its associated chemicals. Studies were selected based on their relevance to RSV's pharmacokinetic changes, therapeutic efficacy, and disease-specific applications.
Results
We evaluated 162 studies in their totality, including preclinical trials, individual clinical trials, and patent investigations. The findings suggest that RSV may be beneficial in the management of chronic problems, including diabetes, cardiovascular disease, neurological disorders, Non-Alcoholic Fatty Liver Disease (NAFLD), inflammatory skin conditions, and some malignancies. Nanocarriers, liposomes, structural derivatives, and dietary preparations are new ways to deliver drugs that have been proven to be more effective. Clinical investigations substantiate its benefits in glycemic control, vascular function, weight loss, and modulation of inflammation. Specific outcomes demonstrate variability among diverse populations and treatment protocols.
Discussion
The review stresses how RSV-based drugs are still getting better, especially with the addition of boronic acid analogues, combination formulations (such as those that include curcumin or flavonoids), and nanotechnology systems for administration. These groundbreaking ideas could improve stability, bioavailability, and function that is unique to certain diseases. Patent data show that RSV is more common in nutritional supplements, cosmetics, and drugs that are said to treat cancer, skin problems, digestive problems, and early aging. But there are still issues with dose optimization, differences among people, and regulatory constraints.
Conclusion
RSV and its compounds provide several therapeutic challenges while potentially facilitating holistic illness management. Their reliability, low cost, and diverse biological activities suggest significant promise for improving future healthcare. In addition, the growing number of patents and available clinical data further support their potential. However, continued research is needed to develop novel strategies for their field application and to better understand the relationship between their structure and biological activity.
1. INTRODUCTION
Resveratrol (RSV) is a polyphenolic molecule that occurs naturally and is mostly found in grapes, blueberries, peanuts, and wine. RSV (trans-3,5,4′-trihydroxystilbene) is a stilbene phenolic molecule that was first found in the root of white hellebore (Veratrum grandiflorum) in 1940 and then again in Vitis vinifera grapevines in 1976 [1]. Trans RSV is a phytoalexin that grapes produce to protect them from fungi like Botrytis cinerea [2]. The “French Paradox” phenomenon, which demonstrates superior heart health outcomes, has been linked to possible health benefits [3].
Most studies, both in vitro and in humans, show that RSV has several preventive effects against diabetes [4]. Several animal studies have shown that RSV is a strong antioxidant that can help protect against a number of diseases, including cancer, diabetes, and heart disease [5-8].
Several investigations have shown that RSV can help prevent diabetes and related problems. These effects include low blood sugar, starting the Sirtuin-1-AMP-activated protein kinase (SIRT-1-AMPK) signaling pathway, starting autophagy, and other molecular mechanisms that are said to lower the risk of diabetes and heart problems [9-11]. Its clinical use has been limited by problems such as low bioavailability, fast metabolism, and volatility. Researchers are working on new ways to distribute RSV that will make it more soluble, stable, and effective. These include formulations based on nanotechnology. This study explores the therapeutic role of RSV in several pharmaceutical indications, focusing on its biochemical targets and mechanisms of action, as well as approaches to enhance its clinical benefit [12].
2. METHOD
2.1. Literature Search Methodology
A systematic literature search was conducted across databases, including PubMed, Scopus, and Web of Science, along with Google Scholar, patent databases, and clinical trial registries from 2000 to 2025. PubMed/ MEDLINE, Google Scholar, Google Patents, and Clinical Trials.gov were the main databases that were explored to obtain the relevant information. As our focus was on patents and clinical trials of RSV conducted over the last two decades, studies and models evaluating its efficacy in vivo and in vitro were examined. The following keyword combinations, “RSV activities,” “inflammatory,” “RSV diabetes,” “RSV patents,” “RSV anti-inflammatory,” “RSV-controlled trials,” “RSV neurodegenerative,” “RSV pharmacokinetics,” “RSV safety concerns,” and “RSV limitations,” were used to extract the data systematically.
The PRISMA 2020 guidelines were followed for study identification, screening, eligibility assessment, and inclusion (Fig. 1). Duplicate records were removed, and studies were screened based on predefined inclusion and exclusion criteria. For this review, studies, patents, and clinical trials published between 2000 and 2025 focusing on RSV or its derivatives with biomedical and pharmacological relevance were included. Only peer-reviewed articles, granted or published patents, and registered trials available in English were considered. Studies were excluded if they were in a language other than English and lacked translation, or if they were abstracts, editorials, or non-peer-reviewed reports. Records unrelated to therapeutic applications, duplicates, or those lacking sufficient methodological details were also excluded. Finally, 162 studies were included for the final work, comprising clinical trials and patent data.
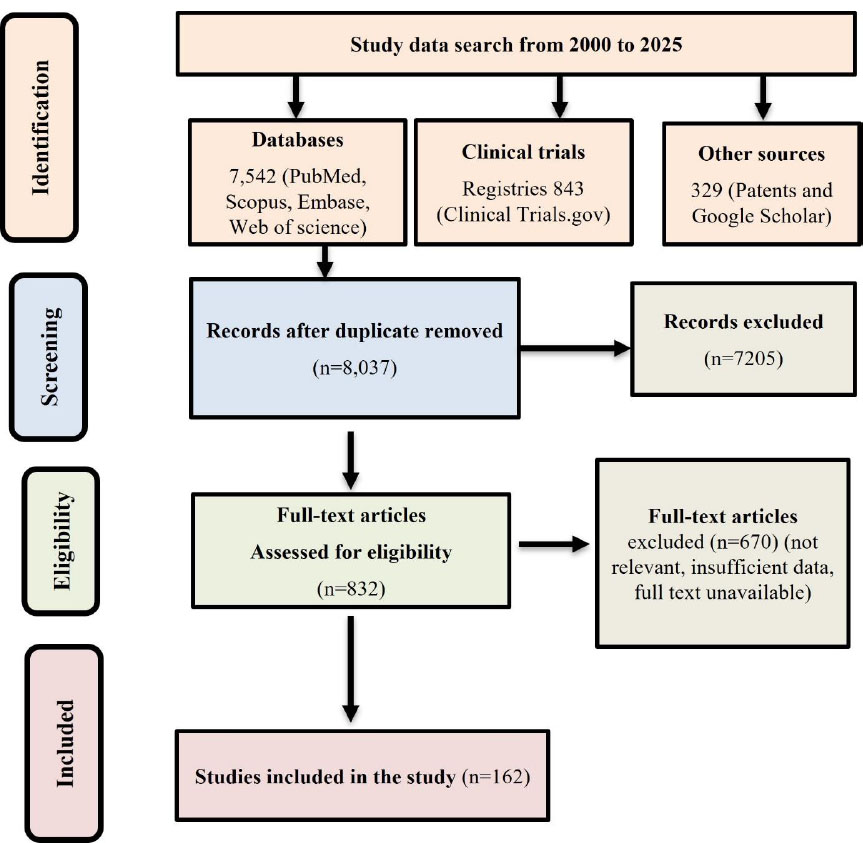
PRISMA 2020 flow diagram of resveratrol study selection (2000–2025), including database, patent, and clinical trial records.
3. RESULTS
3.1. Use of Resveratrol and its Derivatives for Therapeutic Purposes
RSV is a compound that interacts with a variety of targets and can therefore be used as a scaffold to produce stronger reactions with greater mechanistic stringency [13]. In various previous studies, the compound was tested in vitro for its capacity to activate quinone reductase 1 (QR1), inhibit quinone reductase 2 (QR2), produce nitric oxide (NO), obstruct the action of enzyme aromatase, transcription factor protein (NFkB Nuclear factor kappa-light-chain-enhancer of activated B cells), and tissue plasminogen activator (TPA)-induced ornithine decarboxylase (ODC). It was also assessed for the anti-inflammatory effects via cyclooxygenase (COX 1 and 2), its ability to scavenge 2,2-diphenyl-1-picrylhydrazyl free radicals, and its interaction with estrogen receptors [14, 15]. The RSV also demonstrated its effect at all three stages of carcinogenesis, i.e., initiation, promotion, and progression. Additionally, it showed a notable reduction in inflammation in the rat paw model [16]. The RSV can block the transcription factor protein NFκB (nuclear transcription factor), which controls genes associated with inflammatory processes, cellular protection, and cancer formation.The expression of proinflammatory cytokines, including Tumor Necrosis Factor (TNF-α) and interleukins (IL-1, IL-6, and IL-8), is suppressed along with inflammatory mediators such as cyclooxygenase and inducible nitric oxide synthase (5-LOX, COX-2, and iNOS) [17]. Among the numerous ways that RSV and its derivatives function are autophagy, prevention of tumor invasion, promotion of apoptosis, inhibition of angiogenesis, regulation of cell cycle progression, prevention of inflammation, NFkB regulation, and Reactive Oxygen Species (ROS) scavenging, as represented in Fig. (2) below [18].
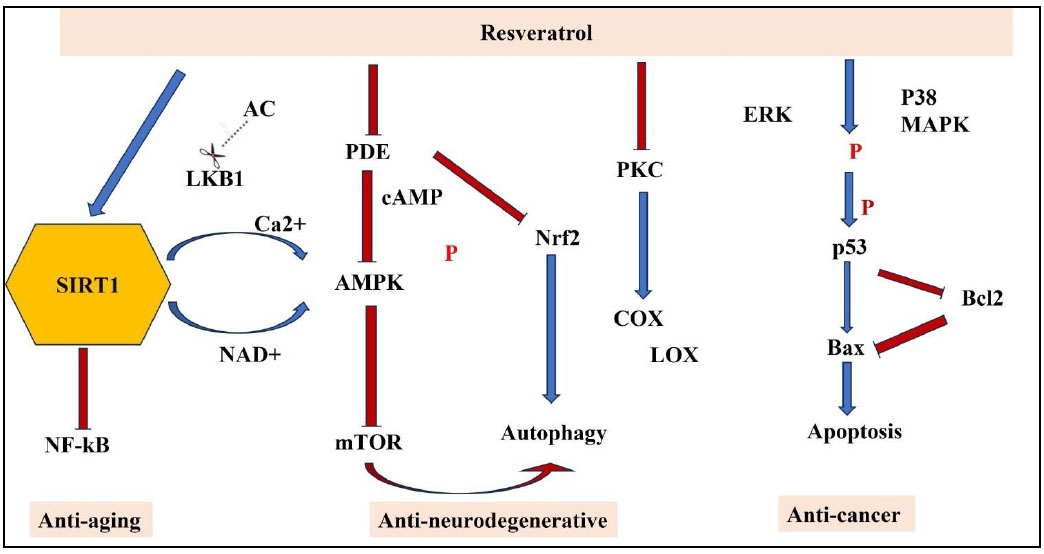
Molecular pathways of resveratrol in anti-aging, neuroprotection, and anti-cancer pathways.
3.2. Anticancer Activity of Resveratrol and its Derivatives
In several animal models, RSV has demonstrated a significant cancer-preventive effect, especially in lowering the incidence of chemically induced skin, breast, liver, gastric, colon, prostate, and neuroblastoma cancers. However, its effectiveness varies by model, with some studies showing no significant effect, such as in spontaneous polyp development, esophageal carcinoma, and certain colon and lung cancer models. These variations demonstrate the importance of factors like cancer type, dose, exposure duration, and experimental design in assessing the chemopreventive potential of RSV [19, 20].
In vitro studies indicate that RSV and its derivatives may be used to prevent and treat cancer. Carcinogenesis progresses over an extended period through successive stages encompassing physiological, cellular, genetic, and molecular transformations, as well as biomarker alterations associated with pharmacological treatments. RSV plays a significant role in regulating many pathways within cancer cells. It can affect how cells grow, cause cells to die, or control how genes are expressed and how hormones interact (Figs. 3 and 4). It also inhibits angiogenesis, reduces inflammation, normalizes redox signaling, and prevents tumor cells from growing. These traits make it a potential therapeutic agent for cancer [21]. RSV has the potential to be an adjunct in cancer therapy by augmenting the effectiveness of chemotherapeutic agents and protecting normal tissues from genetic injury. Combination therapies might benefit from their ability to alter tumor response to treatment and reduce treatment-related harm. Recent advancements, including nanoparticle carriers, have enhanced its therapeutic potential despite its limited bioavailability. More studies are needed to make it more useful for treating cancer [22]. Continuous health monitoring demonstrates that cancer is quite common, which means that new, effective treatments are needed. Plant-based compounds show a lot of potential as medicines because they can affect how cancer works. This evaluation also examines patents covering established plant-derived pharmaceuticals, such as paclitaxel and irinotecan, as well as novel candidates, such as betulinic acid, RSV, and roburic acid [23]. Cancer is an aggressive disease that often requires multiple treatment approaches, many of which are associated with significant side effects. Resveratrol (RSV) shows promise as an inhibitor of cancer progression with minimal adverse effects, despite challenges such as poor bioavailability. Nano-formulations improve RSV’s solubility, enhance its accessibility, and increase its anticancer efficacy. Cancer metastasis remains a huge problem because most drugs do not work well to treat it. RSV has shown promise in preclinical studies for preventing metastasis; nonetheless, progress in therapy remains limited [24].
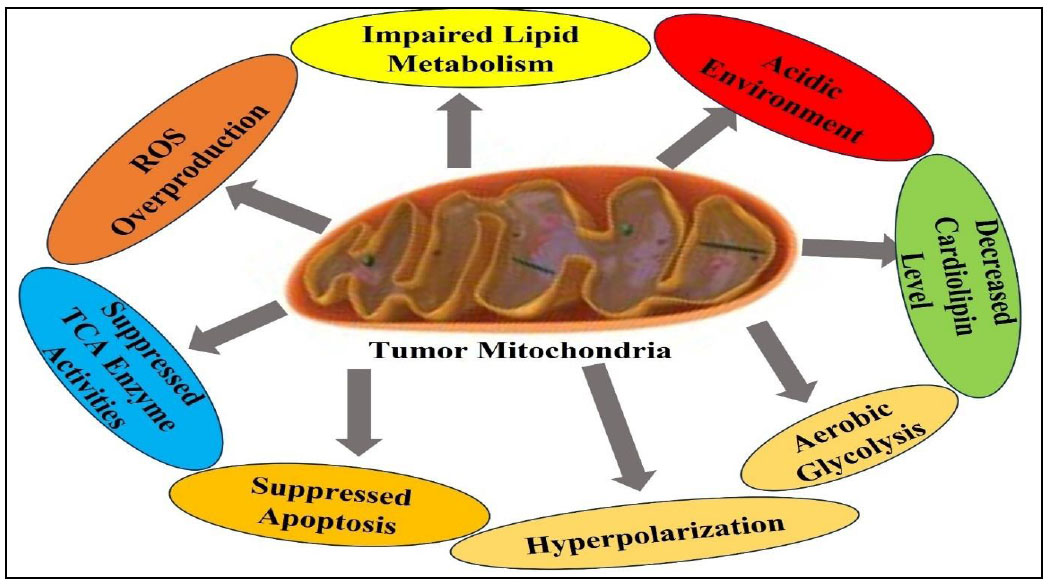
Mitochondrial alterations in tumor cells.
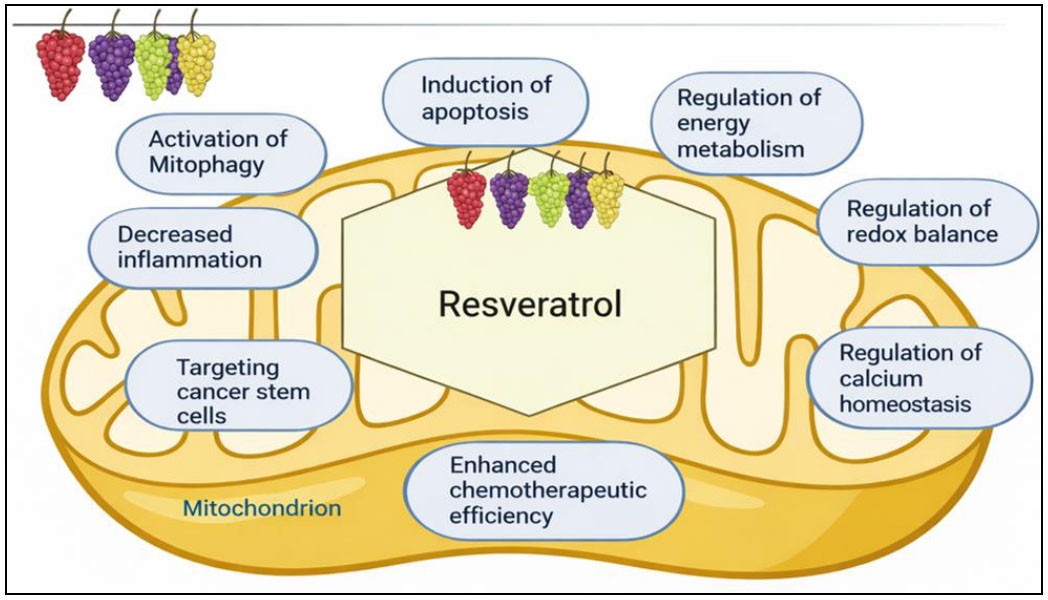
Mitochondrial targets of resveratrol in cancer therapy.
Using boronic acid, researchers developed chalcone analogues of combretastatin A-4. Studies have shown that these compounds are remarkably potent at suppressing carcinoma cells (IC50: 10–200 nM) in 16 different human cancer cell lines. Boronic acid compounds also inhibit proteasome activity, slowing the growth of cancer cells and reducing inflammation [25, 26]. Patented cis- and trans-boronic acid RSV derivatives prove more destructive to estrogen-dependent MCF7 cells than other types of boronic acid. The trans analogue blocks cells in the G1 phase. These derivatives are notably safer and function effectively against cancer cells resistant to multiple medications. They were more effective at combating cancer when coupled with flavopiridol [27]. These results indicate RSV derivatives with boronic acid could be very effective at treating breast cancer in inventive approaches.
Benign Prostate Hyperplasia (BPH) is a prevalent condition in older men, and the most efficient strategy to treat it depends on the severity [28]. Research concludes that RSV and pterostilbene hinder tumors from developing, invading, and spreading in prostate cancer xenografts. This is accomplished by targeting metastasis-associated protein 1 (MTA1), which is a key factor in the growth of tumors [29]. Also, RSV analogues, including trimethoxy-RSV and piceatannol, have shown strong antiproliferative effects in prostate cancer cells and chemopreventive effects in xenograft experiments, which shows their potential could be valuable as therapeutics [30].
A patent published by Leheste and Torres is about formulations for addressing benign prostate hypertrophy, a condition in which RSV has been combined with omega-3 fatty acids, lycopene, or other SIRT1 and Peroxisome Proliferator-Activated Receptor-γ Coactivator 1-alpha (PGC-1α) activators [31]. Research indicates that RSV alone has therapeutic benefits for BPH. In animal models, RSV and trimethoxystilbene reversed testosterone-induced effects similar to finasteride [32]. After 48 hours of exposure to 50 µM RSV, approximately 10% of genes linked to apoptosis were significantly downregulated in normal human prostate endothelial cells [33]. The decrease in prostate weight was associated with a reduction in mitotic activity rather than an increase in apoptotic processes. Formulations of RSV or RSV derivatives may be administered via oral, intravenous, intramuscular (nanoparticle-based), sublingual, or rectal routes, with oral supplements in tablet, liquid, or capsule form.
RSV can also be delivered via probiotics, using enteric bacteria engineered to produce the compound. Additionally, modified RSV forms, where one or more hydroxyl groups are substituted with alkyl, alkenyl, or alkynyl groups, have been explored as potential treatments for type 2 diabetes [34].
Rosacea, an inflammatory skin condition linked to oxidative stress, is characterized by increased cell proliferation. Activating Sirtuin 1 (SIRT1) has been shown to reduce the intricate signaling mechanisms underlying acne, rosacea, and androgenic alopecia, particularly through Androgen Receptor (AR)-mediated growth factor signaling. Advances in understanding nuclear signaling pathways have paved the way for novel treatments targeting diseases linked to androgens, like alopecia, rosacea, and acne vulgaris [35].
3.3. Anti-Aging and Hypolipidemic Properties of Resveratrol and its Derivatives
Recent results from research on animals offer hope for the use of RSV in the management and prevention of metabolic diseases, such as diabetes and obesity. These disorders are quite common, especially in Western nations, and their prevalence is still rising. Obesity is a global epidemic characterized by an increase in adipose tissue mass caused by the production of new fat cells. Adipogenesis is the process by which fat or fatty tissue grows. Overweight and obese people are more likely to suffer from psychological issues such as low self-esteem, eating disorders, and depressive disorders, as well as chronic diseases. The World Health Organization (WHO) reports that over 1 billion adults fall into the overweight category, and at least 300 million are considered clinically obese. The World Health Organization recognizes obesity as one of the foremost contributors to preventable mortality on a global scale [19].
Research employing murine models suggests that RSV would not only alleviate specific adverse effects associated with a calorie-rich diet but also confer benefits similar to those observed with caloric restriction [36]. Interestingly, RSV can mimic calorie restriction because dietary restriction is thought to help prevent several age-related diseases, including insulin resistance, type 2 diabetes, dyslipidemia, and cancer [37]. Several studies show that SIRT1, a NAD+-dependent protein deacetylase, is critical for RSV's physiological effects [38]. The findings further corroborate the critical role of SIRT1 activation in the mechanism of action of RSV, demonstrating that synthetic SIRT1 activators elicit effects analogous to those of RSV [39]. Caloric restriction is also known to augment the expression of the SIRT1 enzyme [40].
Peroxisome Proliferator-Activated Receptors (PPARs) function as ligand-activated transcription factors belonging to the nuclear receptor family. Investigations have demonstrated that fenofibrate and RSV confer neuroprotection against stroke in murine models via a PPARα-dependent pathway. Furthermore, the research indicates that Vaticanol C, a tetramer of RSV, activates both PPARα and PPARβ/δ in vitro and in vivo, potentially providing protection against diseases associated with lifestyle factors [41]. A patent was awarded in 2012 for the discovery of pterostilbene, which acts as an agonist at PPARα receptors [42]. A composition containing the RSV derivative pterostilbene and anthocyanins inhibits adipogenesis, making it effective for weight loss or obesity treatment.
An important risk factor for several human diseases is aging. Increased production of free fatty acids and cholesterol in the liver and skeletal muscle is a sign of lipid homeostasis disruption, a major metabolic change linked to aging. According to recent studies, the possible anti-aging gene SIRT1 may limit lipogenesis by preventing the expression of genes controlled by Sterol Regulatory Element-Binding Proteins (SREBPs) [43].
SREBP-targeted gene expression inhibitors are in great demand. The patent describes boron-containing analogues based on RSV that can be used to treat cancer and dyslipidemias. By inhibiting SREBP-targeted gene expression in both cultured cells and in vivo, the patent identifies boron-containing compounds that prevent the manufacture of fatty acids and cholesterol. Another invention, described by Hull Christopher in 2009, uses RSV topically to lower body fat percentage [44]. Research has shown that Caloric Restriction (CR) can delay age-related changes and increase both average and maximum lifespan in various organisms, including yeast and mammals. It has been demonstrated that CR in mammals can postpone various age-related illnesses, such as diabetes, cancer, atherosclerosis, heart disease, and neurological problems [45]. Oxidative stress and inflammation are induced by a diet rich in lipids and carbohydrates. It is reported that only a single dose of RSV and other grape polyphenols markedly increased the mRNA expression of the glutathione S-transferase-p1 and NAD(P)H dehydrogenase [quinone]1 genes in healthy individuals who had eaten a high-fat, high-carbohydrate diet. The patent covers compositions of stilbene polyphenol derivatives intended to prevent and treat a range of age-related diseases. Additionally, it outlines the procedure for formulating such compositions and highlights their applications in therapeutics, cosmetics, and dietetics [46].
3.4. Anti-Inflammatory Properties of Resveratrol and Related Compounds
Since chronic inflammation has been linked to several malignancies, inflammation inhibition holds promise as a preventative and therapeutic approach [47]. RSV efficiently suppresses IkB kinase activity (IKK), which in turn decreases COX-2 enzyme expression and activation of NF-kB in mouse skin treated with TPA [48].
Intestinal Bowel Disease (IBD) is quite frequent in humans, and certain factors like increased levels of free radicals, pro-inflammatory cytokines, and infiltration of neutrophils are associated with its etiopathogenesis. Anti-inflammatory properties of RSV have been demonstrated [49]. RSV markedly reduced the incidence of colon damage, neutrophil infiltration, and cytokine concentration at the dose of 5 to 10 mg/kg/day in an animal (rat) model of colon injury. As per the Martin et al. study, the RSV molecule reduced COX-2 expression, lowered oxidative stress, and accelerated apoptosis, but did not correct elevated PGE2 levels in rats. These results imply that RSV reduces oxidative damage and triggers apoptosis in rats during the initial phases of colonic inflammation [50].
Acne is a prevalent inflammatory condition affecting more than 85% of teenagers, resulting in serious complications. For minor acne, topical therapies are the initial treatment option. For moderate to severe cases, they are used as an adjuvant therapy. However, most topical therapies have adverse effects such as dryness, redness, and irritation, which may reduce patient compliance [51]. Many factors, including hyperproliferation, raised testosterone levels, increased sebum production, and the creation of proinflammatory mediators, contribute to the etiology of acne [52]. RSV is reported to be readily absorbed through the skin, and hence it has been found to have anti-inflammatory action on topical application in psoriasis [53]. An invention of the University of California, San Diego, UCSD (2014), a topical formulation is described that contains therapeutically effective amounts of RSV and/or its derivatives, either by itself or in conjunction with effective concentrations of benzoyl peroxide and complementary substances like retinoids, antioxidants, antimicrobials like antifungals, etc. Additionally, the composition might contain emulsifiers, surfactants, preservatives, and solubilizers to treat viral and inflammatory skin problems [54]. This innovation offers a way to prevent, treat, and improve the appearance of acne and other infectious or inflammatory skin conditions. Oral formulations can be tablets, syrups, liquid solutions, powders, or gels, whereas topical formulations can be made as creams, ointments, lotions, gels, foams, solutions, or cleansers.
Active RSV derivatives encompass compounds such as cis-3,4’,5-trihydrostilbene, 3,4-dihydroxy-4’-methoxystilbene, 3,5-’dimethoxy-4’-hydroxystilbene’ hydroxystilbene, 3,4’,5-trimethoxystilbene, α,β-dihydroxy-3,4’,5-trihydroxystilbene, cis-piceid, trans-piceid, pterostilbene, ε-viniferin, α-viniferin, δ-viniferin, RSV dimers, 5,4’-dihydroxy-3-O-methoxystilbene, and 3,4-dihydroxy-4’-O-methoxystilbene, among others, including both known and potential future derivatives. RSV, recognized for its antioxidant properties, may be combined with other antioxidants like vitamin C, α-tocopherol, lipoic acids, glutathione, ubiquinol, and polyphenols containing herbal extracts from green tea and coffee berries. Additionally, the composition includes benzoyl peroxide, benzoic acid, and peroxides such as H2O2, which have microbial-killing properties [54]. Although RSV has numerous beneficial properties, no studies have previously assessed its use in treating acne vulgaris.
In the year 2011, a pilot study in Italy, detailed in an abstract, demonstrated its effectiveness in gel form for this condition [55]. Moreover, only one supplementary in vitro study has demonstrated that RSV exhibits antimicrobial, anti-inflammatory, hormonal, and wound-healing properties. These traits help fight the mediator that causes acne, resulting in RSV and its analogues good choices for treating acne and other inflammatory skin conditions, such as rosacea, seborrheic dermatitis, folliculitis, impetigo, ringworm, yeast infections, and onychomycosis [56].
The patent includes information that RSV derivatives can reduce inflammation and suppress the immune system. These compounds exhibit minimal cytotoxicity and suppress inflammation and nitric oxide (NO) synthesis. The composition aimed at the prevention or treatment of inflammatory disorders consists of molecules resulting from the structural modification of RSV, in which the hydroxy group has been substituted with phenyl rings at ortho, meta, or para positions. RSV derivatives are also introduced into functional foods as food additives to prevent or treat inflammatory conditions [15].
Allen's (2015) patent describes a similar invention for treating inflammation in periodontal tissues. Periodontal disease often begins with oxidative damage to a small number of oral cells, which leads to inflammation. Oxidative damage to soft oral tissues can occur from numerous sources, including chemical or biochemical exposure, the use of tooth-whitening agents, or excessive alcohol consumption. Such types of damage can cause inflammation, periodontal disease, heart problems, or increase the risk of tumors in the buccal region [57].
Localized surgical procedures or antibiotics are often used to treat periodontal disease in advanced cases. One of the inventions demonstrates the significance of antioxidants in treating periodontal disease, indicating a clear need for effective treatments. The other issue is figuring out the most effective way to convey it. Antioxidants consumed through food or oral supplements exert only a limited effect on periodontal disease, as they are distributed throughout the body, leaving minimal availability for the gingival tissue. The invention addresses this limitation by describing antioxidant formulations for topical application, designed to treat and prevent periodontal disease. The recommended therapy comprises an oral antioxidant formulation containing a cinnamic acid derivative and two additional antioxidants, including tetrahydrocurcuminoids and RSV [57].
If bacterial plaque spreads below the gum line, gingivitis can progress to periodontitis. As periodontal disease progresses, it may lead to deeper pockets, damage to the bone or gingiva, and destruction of the periodontal ligament, a soft tissue that connects the tooth root to the bone and holds teeth in place. The antioxidant mixtures in this invention can assist in mitigating apoptosis, promoting cell growth, or facilitating easier relocation of periodontal ligament cells. These compositions can also be administered to animals, with potential applications in veterinary care. Delivery methods for these antioxidant formulations may include incorporation into coatings, chew toys, feed, or treats tailored to the veterinary market. Since animals are exposed to different environmental sources of oxidative stress, antioxidant compositions may be specifically designed for veterinary use [58].
3.5. Resveratrol and its Derivatives' Activity in Neurodegenerative Disease
The development and origin of neurodegenerative diseases remain poorly understood, leaving these conditions without cures. Age is the principal risk factor linked to neurodegenerative disorders. In Alzheimer's Disease (AD), the aberrant cleavage of the beta-amyloid peptide leads to the generation of Aβ protein, which is insufficiently eliminated by standard physiological mechanisms. Soluble beta-amyloid oligomers exhibit significant neurotoxicity and aggregate as plaques inside the brain tissue, resulting in localized neuronal damage and eliciting an inflammatory response from glial cells like microglia and macrophages [59, 60]. The glial cells secrete inflammatory cytokines and oxygen-free radicals, which further damage nearby neuronal tissue and worsen the progression of AD [61].
Since RSV affects multiple pathways involved in the pathophysiology of Alzheimer's disease, it has been proposed as a possible treatment for the condition. It has been demonstrated that RSV lowers beta-amyloid levels in brain tissue by encouraging its intracellular degradation via a proteasome-mediated mechanism [62, 63]. Secondly, by activating protein kinase C, it exhibits a neuroprotective effect. Thirdly, certain theories regarding Alzheimer's disease suggest that oxidation may be triggered by elevated levels of heavy metals in the brain, with RSV functioning as a copper-chelating agent [64]. The processes of aging and Alzheimer's disease are closely linked and can be affected by calorie restriction mimetics, with SIRT1 protein acting as the primary mediator. RSV is believed to promote the activation of SIRT1 [65]. Additionally, RSV is a well-known antioxidant that has been shown to protect cultured hippocampal cells from NO-induced neurotoxicity [66]. The bioavailability of RSV has been well investigated. It is highly prone to glucuronidation in the colon and liver, resulting in significantly diminished oral bioavailability. This has caused people to worry about the possible health benefits of eating RSV [48].
Dimauro's (2013) invention describes pharmaceuticals as hybrid compounds that amalgamate RSV and curcumin with various other natural polyphenols. In animal studies, curcumin diminished beta-amyloid levels by 43% when included in the diet. It seems to help reduce the buildup of beta-amyloid [67]. In addition to its positive impact on beta-amyloid levels, it also had strong antioxidant properties and inhibited the expression of pro-inflammatory cytokines [68].
Another group of researchers proposed a similar concept, namely the use of a combination of RSV and piceatannol derivatives to prevent or treat conditions associated with elevated serum IL-6 levels. Taking a specific ERβ agonist, like piceatannol, can help combat certain ailments from getting serious. This innovation is primarily intended to treat chronic inflammatory diseases, migraines, anxiety, and depression [69]. The primary causes of brain disorders, such as depression and migraine, include the release of proinflammatory cytokines and the subsequent activation of NF-kB.
Since estrogen is essential for regulating mental function, a lack of it leads to increased levels of cytokine receptors. The ERβ receptor is the main mediator of estrogen's anxiolytic and depressive effects. In comparison to wild-type mice, ovariectomized ERβ-knockout animals showed markedly lower levels of dopamine and serotonin and higher levels of anxiety. Conversely, ERα seems to act as a mediator of elevated anxiety. In female mice, anxiety behavior and serotonin levels are impacted when ERβ is not functional [70].
The research published by Rimando outlines the anti-anxiety activities of pterostilbene, a natural analogue of RSV. The researcher has demonstrated the chemopreventive and antioxidant properties of pterostilbene. In a mouse model, when pterostilbene was administered via the oral route, it exhibited anti-anxiety-like effects similar to the standard drug diazepam, with no impact on locomotor activity at any dose. Following a single oral dosage, pterostilbene was found in both serum and brain tissue, suggesting that the substance has an anxiolytic effect and can penetrate the blood-brain barrier. RSV, on the other hand, did not show any anxiolytic effects [71].
3.6. Nutraceutical Compositions Comprising Resveratrol and its Derivatives
Dr. Stephen DeFelice coined the term “nutraceutical,” which refers to a food that offers medical or health benefits, including the prevention or treatment of diseases [72]. The investigation of nutritional supplements with therapeutic applications has evolved into an intriguing area of research, leading to the development of diverse products, including nutritional bars, energy drinks, cereals, and other items enhanced with nutraceutical ingredients.
The pharmaceutical biotechnology industry has recognized the growing preference for nutraceuticals over traditional pharmaceuticals, primarily because they can serve large populations while delivering favorable therapeutic benefits [73]. Crystalline drug forms offer enhanced physicochemical stability while preserving the quality of the active pharmaceutical ingredient [74].
In his patent, Espin de Gea (2012) outlines a combination of RSV derivatives, including glucopyranoside derivatives. These derivatives are meant to treat or alleviate intestinal inflammatory diseases. These derivatives can be incorporated into various compositions such as food, dietary supplements, functional foods, or nutraceuticals. By modifying the structure of the RSV, its active component bypasses Phase II metabolism enzymes, thereby ensuring it effectively circulates to the intestine. The researchers used the C57BL/6 mouse model for colitis and found that the levels of MIP (Macrophage Inflammatory Protein), pro-inflammatory cytokines, and prostaglandin E2, which are all mediators of inflammation, were significantly decreased [75].
The formulation might be useful for other inflammatory diseases as well as treating intestinal inflammatory diseases. These include cancer, cardiovascular disease, and neurodegenerative disorders. Synthetic cyclodextrins with great water affinity have been used in RSV-based formulations [76]. In patent Souto (2014), an approach to produce a water-soluble cyclodextrin complex with methylated and acetylated derivatives of RSV is described. This is meant to increase the drug's oral bioavailability. The derivatives were dissolved in rice bran oil, enriching the formulation with the effects of rice bran constituents. This formulation, designed as a nutraceutical product, enhances the therapeutic efficacy of RSV through the synergistic properties of rice bran oil, which is abundant in tocopherols and phytosteroids. In Wistar rats, the administration of an RSV-derivative complex in rice bran oil resulted in a substantial decrease in chronic inflammation and associated depressed behaviours [77].
Recent patents have explored new combinations of RSV and pterostilbene for disease treatment and nutraceutical applications. Patent US8399712B2 describes pterostilbene (the dimethoxy derivative of RSV) cocrystals with enhanced bioavailability, as well as improved anti-cancer, antioxidant, and anti-inflammatory properties. This cocrystal formulation may also help manage hyperglycemia and hypercholesterolemia [78]. Many other patents have also been published on the structural modification and substitute analog preparation of RSV [79, 80]. Another significant patent, WO2009089338A2, addressed methods for delivering RSV and pterostilbene using controlled-release dosage forms, including chewable forms designed to improve absorption through mucosal surfaces. This approach focuses on increasing the bioavailability of these compounds, which may benefit patients with certain cancers and cardiovascular diseases [81].
Studies in the nutraceutical field are focusing on encapsulating RSV using kafirin and milk protein nanoparticles to enhance protection against degradation and facilitate bioavailability. In this study, encapsulation efficiency ranged from 67 to 76%, with the kafirin/casein matrices exhibiting the highest efficiency. These formulations are designed to enhance the stability and bioavailability of RSV in dietary supplements [82]. The Chinese patent CN102939080A pertains to 4-acetoxy-RSV, which is a modified version of the stabilized form of bioactive RSV. It shows that modified RSV has greater bioavailability and greater biological potency in the digestive tract than regular RSV [83]. It also has enhanced bioactive properties compared to regular RSV, including anti-inflammatory and antioxidative effects, cancer chemoprevention, and management of metabolic disorders.
Innovative formulation technologies, along with the combined use of RSV and pterostilbene, show promising and expanding opportunities for these compounds as nutraceuticals in the treatment of multiple disorders. New developments and research are focused on maximizing the therapeutic complexity and effectiveness of these new applications.
Table 1 below outlines the details of the patents of RSV/RSV analogues/their combinations cited in the above paragraphs.
| Patent No. | Filed/Published Year | Active Ingredients/compounds | Current Assignee | Remarks | Reference |
|---|---|---|---|---|---|
| US20120149663A1 | 2010/2012 | Boronic acid derivatives of RSV | Georgetown University | Improved the cytotoxicity in the MCF-7 cell line, induced G1 cells, and had an irreversible antiproliferative effect against the MDR cell line, treating cancer. | [20] |
| US20100048577A1 | 2009/2010 | 3,5’-dimethoxy-4-stilbenol | New York Institute of Technology | Treatment of BPH (Benign Prostate Hyperplasia) using RSV | [31] |
| CN102050759A | 2010/2011 | RSV amide derivative | Jiangsu Tiansheng Pharmaceutical Co., Ltd. | Antineoplastic drug | [84] |
| WO2011039175A1 | 2010/2011 | Piceatannol and RSV | protection from skin cancer in smokers | [85] | |
| CN103547256A | 2012/2014 | RSV derivatives have various positions around the central core benzene rings. | The University of California, San Diego, UCSD | Skin conditions caused by inflammation and infection. | [54] |
| US20120309699A1 | 2010/2012 | RSV trans-spliced analogues | Consejo Superior De Investigaciones Cientificas (Csic) | The compound has anti-inflammatory effects in the intestine. | [75] |
| AU2009319881C1 | 2009/2015 | Stilbene derivative, antioxidant composition includes cinnamic acid derivatives | Perio Sciences LLC | Diseases of the oral cavity, including periodontal inflammation | [57] |
| US8350093B2 | 2010/2013 | hybrid molecule: methylated curcumin and RSV. | DePuy Spine LLC, DePuy Synthes Products | Utilized in the treatment of Alzheimer’s disease. | [67] |
| US8268792B2 | 2006/2012 | A blend of RSV and piceatannol precursors along with their stereoisomeric variations. | Individual | Conditions linked to elevated IL-6 serum levels include depression, anxiety, migraines, chronic inflammatory diseases, tumorigenesis prevention, and prostate cancer treatment. | [69] |
| WO2012154956A2 | 2016/2016 | pterostilbene | The United States of America, as represented by the Secretary of Agriculture, University of Mississippi | Anxiolytic effect | [86] |
| WO2012156275A1 | 2016/2016 | RSV | Difass International S.R.L. | Pruritus | [87] |
| US20120058088A1 | 2014/2014 | A phenolic antioxidant and metal chelator, trans-RSV, demonstrates significant therapeutic potential for vitamin D. | Resveratrol Partners LLC | To enhance cell transformation therapies and address conditions like macular degeneration or dystrophy. | [88] |
| US8992603B2 | 2012/2015 | Polyphenolic compounds such as RSV and quercetin. | Nanocopoeia, Inc., Louisiana State University Health Sciences Center Office of Research | Devices with coatings minimize smooth muscle cell proliferation and platelet aggregation, reducing the risk of atherosclerosis, stenosis, and clotting-related diseases. | [89] |
| US20120165280A1 | 2011/2015 | Stilbenoid analogues | BioVentures LLC | To promote better blood circulation. | [90] |
| US20100310615A1 | 2008/2010 | Composition of piceatannol, RSV, etc. | Caudalie | In therapies targeting aging | [46] |
| US20090326000A1 | 2008/2009 | The combination of RSV, its esters, amides, and structural analogues | Individual | Managing or combating obesity | [44] |
| WO2012112670A1 | 2012/2012 | Boron-containing derivatives of RSV | Albert Einstein College of Medicine of Yeshiva University, The Texas A&M University System | Targeting lipid disorders and cancer | [43] |
| US20120178801A1 | 2011/2012 | A formulation containing one or more stilbene derivatives and anthocyanins | Sami Chemicals and Extracts Ltd. | Applied for weight management or promoting weight reduction | [42] |
| WO2010004256A8 | 31/03/2011 | At least one compound chosen from the group including RSV, tramadol, acetaminophen, xorphanol, etc. | E-Therapeutics Plc | Regulated release therapy for managing depression | [91] |
| US10716815B2 | 2019/2020 | RSV | Seed Health Inc. | employed to treat and prevent cancer and other age-related diseases | [92] |
| US11524043B2 | 2021/2022 | RSV derivatives. | 4Life Patents LLC | Composition including an immune modulator and adaptogens | [93] |
| KR102576222B1 | 2022/2023 | RSV with other derivatives | Korea Institute of Oriental Medicine, Gyeonggi Economic and Scientific Promotion Agency | Composition used for prevention/amelioration or treatment of Alzheimer's disease | [94] |
| WO2023219526A1 | 2022/2023 | Dietary supplement composition with RSV | Individual | Dietary supplement composition for antiviral activity | [95] |
| KR102597760B1 | 2023/2024 | RSV nanoparticles containing ceramide for cosmetic applications | Cosmecca Korea Co., Ltd., A-Biomaterials Co., Ltd. | For skin moisturization, antioxidants, inflammation, etc. | [96] |
3.7. Clinical Trials on Resveratrol from the 20th Century Onwards
RSV naturally occurs in two configurations (cis and trans), with the latter being noteworthy for its health benefits [97]. The trans form demonstrates significantly greater bioavailability than the cis form and is the more stable form abundant in nature [98, 99]. The varied biological functions of RSV have been validated through preclinical and clinical research.
As indicated in Table 2 below, clinical trials have examined the preventive and therapeutic effects of RSV in a number of illnesses and conditions, such as diabetes, obesity, cancer, neurological disorders, cardiovascular diseases (CVDs), and specific bacterial and viral infections [100]. Both clinical and preclinical studies have demonstrated that RSV targets multiple pathways and many signaling molecules. They encompass transcription factor (NF-κB), cytokines, apoptosis-triggering enzymes (caspases), MMPs (matrix metalloproteinases), AMPK (5' AMP-activated protein kinase), and adhesion molecules like VCAM (vascular cell adhesion molecule) and ICAM (intercellular adhesion molecule). Others are the SIRT1 enzyme, TNF-α, PGC-1α (peroxisome proliferator-activated receptor gamma coactivator 1-alpha), and growth factors like IGF-1 (insulin-like growth factor 1), IGFBP-3 (insulin-like growth factor-binding protein 3), and VEGF (vascular endothelial growth factor). RSV also targets the Ras association domain family 1 isoform A (RASSF-1α), pAkt, VEGF, COX-2, nuclear factor erythroid 2-related factor 2 (Nrf2), and Kelch-like ECH-associated protein 1 [101, 102]. Its ability to interact with a variety of targets, such as kinases, receptor proteins, and the aforementioned signaling molecules, contributes to its complex actions [103, 104].
| Disease Type | Target Patient’S Number | Dose Administered / Route / Quantity, Duration | Effect/Biochemical Changes/Outcomes | Ref. |
|---|---|---|---|---|
| T2DM | 66 | 1 g/day for 45 days, orally | Remarkably lowered HbA1c, fasting blood glucose, insulin, and insulin resistance. | [105] |
| 19 | 5 mg/day for 4 weeks, orally 2 times | Improved the insulin sensitivity, reduced oxidative stress and blood glucose concentration, decreased urinary ortho-tyrosine excretion, and increased the pAkt: Akt ratio in platelets | [106] | |
| 43 | 480 mg/day for 4 weeks, orally | Decreased insulin levels and insulin resistance, along with enhanced periodontal health. | [107] | |
| 24 | 50 mg/day, 2 times daily for 60 days, orally | Accelerated healing and a decrease in the size of foot ulcers, and a decline in plasma fibrinogen level. | [108] | |
| 35 | Dietary RSV extract 8 mg/day for 12 months. | Lowered serum levels of inflammatory biomarkers IL-6 and ALP, along with lowered expression of inflammatory cytokines CCL3, IL-1β, and TNF-α. | [109] | |
| 62 | 250 mg/day for 3 months along with oral hypoglycemic agents | Reduced HbA1c levels, systolic blood pressure, total cholesterol, and total protein. | [110] | |
| 36 | 0.75, 150, or 300 mg/weekly of RSV | Improved neurovascular function and cognitive performance in patients with diabetes. | [111] | |
| Obesity | 24 | Oral RSV 1500 mg/day (500 mg × 3 daily) for 4 weeks | No notable changes in insulin sensitivity. | [112] |
| 11 | Oral ResVida® 150 mg/day for 30 days (placebo vs. RSV, 4-week washout). | Decreased sleep and resting metabolic rates, activation of AMPK led to increased SIRT1 and PGC-1α levels, enhanced mitochondrial efficiency, and reduced ALT, IL-6, and TNF-α levels. | [113] | |
| 28 | Oral trans-RSV 75 mg/day for 6 weeks | Enhanced flow-mediated vasodilation (FMD) by ~23% vs placebo (P≈0.02); a single dose after chronic treatment produced greater acute FMD. | [114] | |
| 19 | Single oral doses of 30, 90, and 270 mg (resVida™) vs. placebo | Produced an acute dose-dependent enhanced FMD | [115] | |
| 10 | Oral Resvida® 150 mg/day for 30 days | Decreased glucagon response after meals, no effect on GLP-1 or GIP (fasting/postprandial), but suppressed postprandial glucagon responses. | [116] | |
| 8 | 1000 mg/day, 1st week; 2000 mg/day, 2nd week |
Decreased production of ApoB-100 rate by ~22% and ApoB-48 lipoprotein rate by ~22% | [117] | |
| 11 | Oral 150 mg/day for 30 days | Enhanced adipogenesis and a reduction in adipocyte size. | [118] | |
| 32a | Oral equimolar doses (0.66 mmol) of RES, RSV-Triphosphate (RTP), or Catechin-Rich Grape Seed Extract (CGSE) for 28 days | Markedly decreased oxidative stress. | [119] | |
| 161b | Orlistat (120 mg) and RSV (100 mg), 3 times a day for 6 months | Substantial reduction in body weight, improved BMI, waist circumference, fat mass, triglycerides, leptin, and leptin/adiponectin ratio. | [120] | |
| Overweight | 29b | Dietary RSV for 8 weeks | Notable reductions in fasting and PP plasma glucose, enhanced glyoxalase 1 expression, and reduced plasma methylglyoxal expression. | [121] |
| 32 | 300 and 1000 mg/day, 2 times for 12 weeks |
Lowered blood glucose levels | [122] | |
| Colon cancer | 8 | 80 and 20 mg/day for 14 days (RSV formulation or freeze-dried grape powder) | Increased expression of myc, axinII, and cyclin D1, along with decreased expression of CD133 and LGR5 in normal colonic mucosa. | [123] |
| Colorectal cancer | 9a | 5 g/day for 14 days | Enhanced bioavailability, good tolerability, and raised caspase-3 levels in malignant hepatic tissue with hepatic metastases. | [124] |
| Multiple myeloma | 24a | 5 g/day for 20 days | Caused adverse events in patients; no clinical benefits observed | [125] |
| Breast cancer | 39 | 5 and 50 mg/day 2 times for 12 weeks | Inhibited methylation of RASSF-1α and modulation of PGE2 in breast tissue in breast cancer patients. | [126] |
| BRPC | 14b | 4.4‐35.2 µg/day for 28 days | Well tolerated by patients, insignificantly increased PSADT, unaltered PSA level |
[127] |
| NAFLD | 50 | 500 mg/day for 12 weeks | Decreased ALT levels and hepatic steatosis, improvements in some cardiovascular risk factors, and metabolic markers in NAFLD patients | [128] |
| 60 | 300 mg/day 2 times for 3 months | Decreased levels of glucose, insulin resistance, cholesterol, ALT, and AST; significant reduction in TNF-α, cytokeratin-18 fragment, and fibroblast growth factor, along with an increase in adiponectin levels. | [129] | |
| 20 | 3000 mg/day for 8 weeks | No notable improvements were observed in the features of NAFLD. | [130] | |
| Alzheimer’s disease | 119 | 500 mg/day with a 500 mg increment every 13 weeks, and ending with 1000 mg/day 2 times | No significant clinical benefits observed. | [131] |
| Brain ischemic stroke | 312 | 2.5 mg/kg (max 250 mg) as an infusion bolus | Expanded the narrow clinical therapeutic window, decreased MMP-2 and MMP-9 levels, and enhanced NIHSS scores. | [132] |
| Overweight older | 46 | 200 mg/day for 26 weeks | Enhanced functional connectivity in the adult hippocampus and better memory performance. | [133] |
| Healthy adults | 22 | 250 and 500 mg/week for 3 weeks | Elevated cerebral blood flow in a dose-dependent fashion. | [134] |
| 60b | 500 mg/day for 28 days | No effect on sleep or cerebral blood flow. | [135] | |
| Postmenopausal women | 80 | 75 mg 2 times a day for 14 weeks | Enhanced mood and cognitive function. | [136] |
| Primary CVD | 75b | RSV-rich grape extract capsule (350 mg) daily for 6 months | Marked reduction in cholesterol and Apo-B levels. | [137] |
| 75b | Dietary RSV-rich supplement (8 mg) for 6 months, with double dose for the next 6 months | Reduced levels of hsCRP, TNF-α, and PAI-1. | [138] | |
| Patients with CVD risk | 1000b | Dietary, random | Enhanced good blood lipid profile and lowered fasting blood glucose levels decrease the risk of CVD. | [139] |
| Angina pectoris | 166b | 20 mg/day for 60 days | Reduced hsCRP and brain natriuretic peptide levels. | [140] |
| Myocardial infarction | 40 | 10 mg/day for 3 months | Enhanced left ventricular diastolic and endothelial function, with a reduction in LDL. | [141] |
| Healthy subject | 44b | 400 mg/day for 1 month | Markedly decreased expression of ICAM, VCAM, and IL-8, along with reductions in plasma IFN-γ levels and fasting insulin concentrations. | [142] |
| CKD | 72 | 150 mg/day (low dose) and 450 mg/day (high dose) for 12 weeks | Notable enhancement in renal function, accompanied by substantial improvements in peritoneal angiogenesis markers, including VEGF, Flk-1, Ang-2, Tie-2, and TSP-1. | [143] |
| 20 | 500 mg/day for 4 weeks | No impact on proinflammatory or antioxidant biomarkers. | [144] | |
| Takayasu arteritis | 242 | 250 mg/day for 3 months | Decreased levels of CRP and TNF-α, with improved BVAS scores. | [145] |
| 10 | 5 g/day for 2 days | Increased TNF‐α and increased activation of the NF‐κB pathway | [146] | |
| Healthy subjects | 10 | Dietary RSV (40 mg /day) for 6 weeks |
Reduced plasma concentrations of TNF-α, IL-6, and CRP, along with inhibited expression of JNK1 and IKKβ. | [45] |
| PCOS | 34 | 30 mg/day for 2 months | Substantially lowered testosterone levels and decreased dehydroepiandrosterone sulfate. | [147] |
| Cancer protection activity | 40 | 0.5-5 g/day for 4 weeks | Metabolites (RSV-3-O-sulfate, RSV-4′-O-glucuronide, and RSV-3-O-glucuronide) reached plasma levels/AUC up to 20.3× higher than parent RSV. | [148] |
| Endometrial pain | 42 | 30 mg/day for 2 months | A notable decrease in endometrial pain scores, along with a significant reduction in aromatase and COX-2 expression, has been observed. | [149] |
| Schizophrenia | 19 | 200 mg/day for 4 weeks | Lower triglycerides level and prevent lipid profile damage. | [150] |
| Rhinopharyngitis | 82a, b | Aerosol formulation 2 times a day for 20 days | Marked reduction in rhinorrhea, nasal congestion, cough, fever, and sneezing. | [151] |
| Ulcerative colitis | 50 | 500 mg/day for 6 weeks | A substantial decrease in TNF-α, hsCRP, and NF-κB levels in peripheral blood mononuclear cells, along with a reduced colitis activity index, has been observed. | [152] |
| Sedentary adults | 13 | 500 mg/day 2 times a day for 1 week and 1000 mg/day 2 times a day for the next 3 weeks | No notable effects. | [153] |
| Hypertension | 97a, b | 50 mg/day for 6 months | Lowered blood pressure accompanied by a significant decrease in ALT and γ-GT levels. | [154] |
| Healthy adult | 50 | 500 mg/day for 30 days | A significant decrease in CRP and triglycerides, along with an increase in total antioxidant levels, was observed in smokers. | [155] |
| Acne vulgaris | 20a | RSV-containing gel for 60 days | A decrease in the acne grading system score. | [55] |
Note: a. Formulation/modified RSV, b. Combination study.
Rapid metabolism in the human body hinders the clinical application of RSV, despite its numerous targets. The oral bioavailability of RSV is extremely low, approximately 1%, significantly restricting its therapeutic effectiveness, especially in organs far from the gastrointestinal tract. However, clinical trials have demonstrated that RSV exhibits activity both as a standalone agent and in combination with other compounds. This review highlights the documented clinical effects of RSV, along with its pharmacokinetics, pharmacodynamics, molecular targets, and potential side effects in human participants.
3.8. Completed Clinical Trials on Resveratrol and its Derivatives
RSV, initially isolated and identified in 1939, did not gain significant attention from clinicians and researchers until the 1990s [156-158]. To the best of our knowledge, well-designed clinical trials on RSV have only been published since the early 21st century. Over the past two decades, interest in RSV's clinical applications has grown steadily, as evidenced by the increasing number of annual publications on the topic (Fig. 5). Currently, the PubMed database (http://www.ncbi.nlm.nih.gov/pubmed) lists more than 297 clinical studies. Notably, several RSV clinical trials registered on www.clinicaltrials.gov remain unpublished, even though most have been completed (summarized in Table 3). This section explores the findings from clinical trials reported in published studies.
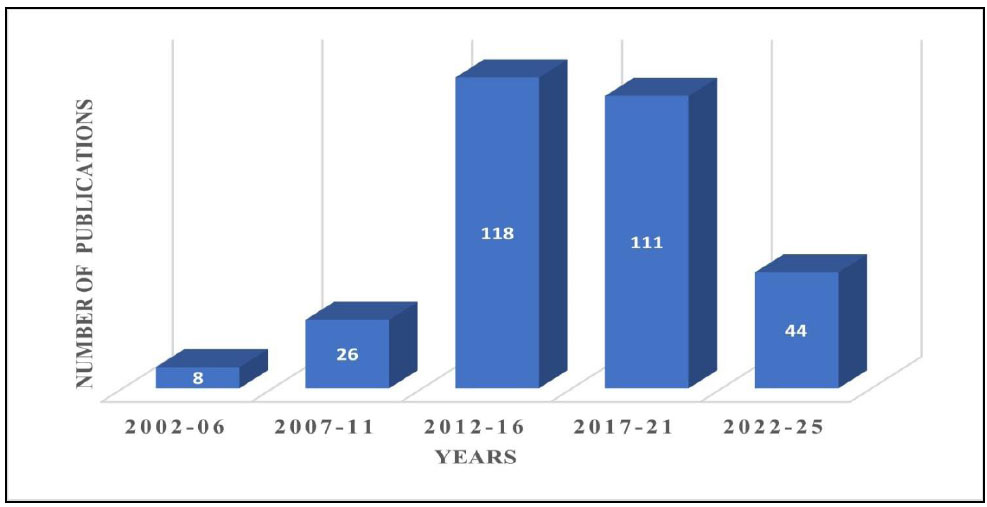
Trends in the number of publications related to the clinical applications of resveratrol from 2002 to 2025.
| Disease Type | Identifier No. | Phase I/II/III/IV | Targeted Patents | Dose, Duration | Sponsor/Principal Investigator Affiliation | Start Date |
|---|---|---|---|---|---|---|
| T2DM | NCT01677611 | I | 10 | 500 mg /day‐3 g/day, 12 weeks |
Kian Peng Goh, KTPH, Singapore | 2008 |
| T2DM | NCT01038089 | - | 20 | 90 mg/day and 270 mg/day, 2 weeks | Joseph A. Vita, MD, Boston University | 2009 |
| T2DM | NCT02549924 | II | 22 | 3 × 500 mg/day, 8 weeks | Martínez‐Abundis E, CUCS, UG, México | 2015 |
| Diabetic nephropathy | NCT02704494 | Early phase I | 60 | 500 mg/day, 3-months | Ranjbar G et al., SUMS, Iran | 2016 |
| Obesity | NCT00998504 | - | 18 | 150 mg/day, 30 days | Silvie Timmers, MUMC, The Netherlands | 2009 |
| Obesity, IS, T2DM | NCT01302639 | - | 18 | Dietary, 3-day | MUMC, The Netherlands | 2011 |
| Obesity, MS, diabetes | NCT00823381 | - | 58 | 75 mg/day, 3 months | Samuel Klein, M.D., Washington University School of Medicine, USA | 2009 |
| Obesity/MS | NCT01150955 | - | 24 | 3 × 500 mg/day, 5 weeks | Jens Otto L. Jorgensen, AUH, Denmark | 2010 |
| Metabolic syndrome X | NCT02114892 | II | 24 | 1500 mg/day, 90 days | Manuel Gonzalez, UG, Mexico | 2012 |
| Colon cancer | NCT00256334 | I | 11 | 20, 80, and 160 mg/day and dietary, 14-day | Randall Holcombe, UC, USA | 2005 |
| CRC | NCT00433576 | I | 20 | Dietary, 8-day | Dean Brenner, UMN, USA | 2006 |
| CVD | NCT01449110 | II | 150 | Dietary, 6 months | Juan Carlos Espin, NRC, Spain | 2009 |
| Heart failure/disease | NCT01185067 | I | 15 | Dietary, 6 weeks | Scott L. Hummel, UM, USA | 2010 |
| Renal insufficiency | NCT02433925 | III | 20 | 500 mg/day, 4 weeks | Universidade Federal Fluminense, Brazil | 2013 |
| CKD | NCT03352895 | - | 36 | 100 mg/day, 1 year | JH Hwang, DTCGH, Taiwan | 2014 |
| Solid tumor | NCT00098969 | I | 40 | Dietary, 1 day | Dean E. Brenner, UMCC, USA | 2004 |
| CVS | NCT03762096 | - | 40 | 2 g/day | Douglas Sawyer, Maine Health, USA | 2018 |
| Knee Osteoarthritis | NCT02905799 | III | 142 | 40 mg/day, 6 months | Christelle Nguyen, MD, PhD, UPD, France | 2017 |
| CKD | NCT03597568 | - | 25 | 400 mg/d, 2 weeks | Diana Jalal, University of Iowa, United States | 2019 |
| Aging, healthy aging, and obesity | NCT05981053 | - | 30 | 150 mg/L/day, 90 days | University of Buenos Aires, Argentina | 2022 |
| Diabetic Neuropathies | NCT05172947 | - | 120 | 500 mg/day, 3 months | Bushra Marouf, PhD, University of Sulaimani, Iraq | 2021 |
| Skin conditions | NCT04456829 | - | 60 | 1 bottle of RSV drink, 2 months | TCI Co., Ltd., Taiwan | 2020 |
| Cognitive change, mood, systemic change | NCT04314739 | - | 100 | 500 mg/d, 4 weeks | Emma L. Wightman, Dr., Northumbria University | 2019 |
Note: (Source: https://www.ncbi.nlm.nih.gov/pubmed/).
3.9. Clinical Trials in Diabetes, Obesity, and Metabolic Syndrome Management
Phytomolecules are frequently used to manage metabolic disorders by shifting the body from a state of metabolic stress to one of balance, potentially improving insulin sensitivity and supporting glycemic control. Among these disorders, diabetes mellitus, commonly referred to as diabetes, is one of the most prevalent worldwide. Its global burden is projected to reach approximately 552 million cases by 2030 [159, 160].
RSV has shown significant potential in addressing various metabolic conditions, including diabetes, obesity, and Metabolic Syndrome (MS) [161]. RSV was provided to Type 2 diabetic patients at a dose of 1 gram per day for 45 days in a clinical trial involving a placebo control group. Glycated haemoglobin (HbA1c), insulin, and fasting blood glucose all significantly decreased, while High-Density Lipoprotein (HDL) cholesterol levels increased following this intervention [105]. In addition, diabetic patients' blood sugar parameters and responsiveness to insulin improved with lower RSV dosages (10 mg per day for 4 weeks). These advantages were linked to increased phosphorylation of protein kinase B (Akt), a crucial component of insulin signalling pathways, and reduced oxidative stress, as indicated by lower urinary ortho-tyrosine levels [106]. A meta-analysis confirmed RSV's positive effects in diabetics, although it found little effect in healthy subjects [162].
According to a different meta-analysis, RSV is more effective at controlling diabetes when given in lower doses. Nevertheless, at least one clinical study found no appreciable metabolic advantages to low-dose RSV treatment [163]. These disparities might result from differences in patient characteristics like age, sex, and body weight, which might impact how RSV behaves pharmacologically in various clinical scenarios. The condition Type 2 Diabetes Mellitus (T2DM) is often linked to various secondary medical issues, especially in elderly patients, necessitating more extensive healthcare as well as disease management [164]. RSV has been identified as a possible medication for managing concurrent illnesses in elderly people with diabetes [165]. For instance, in individuals with T2DM and chronic gum disease, RSV supplemental intake resulted in a notable rise in insulin levels while decreasing insulin resistance, simultaneously improving oral health. However, it exerted negligible effects on fasting blood glucose and triglyceride levels [107]. About 25% of people with diabetes develop diabetic foot ulcers, which progressively can worsen if they are not treated [166, 167]. RSV has demonstrated efficacy in facilitating wound healing and diminishing ulcer size [108]. It is believed that these effects happen when AMP-activated protein kinase (AMPK) is activated. This makes wound sites more vascularized or neovascularized and helps granulation tissue grow [168, 169].
In animal studies, RSV has demonstrated the ability to lower the glycemic index by boosting Glucagon-Like Peptide-1 (GLP-1), which enhances insulin secretion and inhibits glucagon release [170]. Nonetheless, not all clinical studies corroborate its antidiabetic efficacy in humans. In a study with 14 T2DM patients, high-dose RSV (1 g/day for 5 weeks) did not result in significant alterations in glycemic control, body weight, or HbA1c levels relative to a placebo group. Additionally, GLP-1 activity was not elevated in these patients [171].
RSV significantly reduced postprandial glucagon levels independently of GLP-1, indicating that alternative mechanisms may underlie its metabolic advantages. Additionally, RSV may enhance the negative correlation between insulin sensitivity and hypertension, providing further benefits in the management of diabetic complications [106]. In general, RSV could help with long-term secondary problems that come with type 2 diabetes, especially in older people who are more likely to have more than one health problem at a time.
Chronic inflammation is becoming increasingly recognized as a pivotal contributor to the onset of various metabolic disorders, including obesity, Type 2 Diabetes Mellitus (T2DM), Impaired Glucose Tolerance (IGT), and impaired fasting glucose [172, 173]. It is especially involved in the development of insulin resistance [174], with increased concentrations of inflammatory cytokines like TNF-α, IL-1, IL-6, and IL-8 playing a role in this condition. IL-1β has been associated with pancreatic β-cell apoptosis [175]. Grape Extract (GE), a natural source of RSV, has been utilized both independently and in conjunction with RSV to alleviate inflammation. In a one-year clinical investigation, patients with T2DM who received RSV-enriched GE had a considerable decline in IL-6 and alkaline phosphatase (ALP). Those who only got GE saw a drop in ALP only in the first six months [109]. A decrease in ALP is linked to a decrease in inflammation [176, 177]. Additionally, polyphenols such as RSV can modulate inflammatory and metabolic pathways through microRNA regulation [178, 179]. Targeting pro-inflammatory cytokines may therefore be advantageous for patients with prediabetes, a precursor to several chronic diseases [180, 181]. However, results from two short-term clinical trials indicate that high doses of RSV did not significantly lower serum IL-6 levels in T2DM patients, highlighting the necessity for additional research into its anti-inflammatory properties [112, 182].
Hypertension frequently coexists with diabetes, typically resulting from heightened fluid retention and arterial rigidity [183, 184]. Chronic hypercholesterolemia can worsen high blood pressure by narrowing the arteries [185]. RSV is often used with standard diabetes treatments because it is known to be safe at low doses [154]. In one study, 250 mg/day of RSV supplementation in T2DM patients improved HbA1c, systolic blood pressure, and total cholesterol levels [110]. Furthermore, T2DM patients have a higher risk of cognitive decline as their cerebral blood flow is lower. A 75 mg dosage of RSV increased vasodilation in the cerebral arteries, which may have improved the blood flow to the brain [112]. RSV boosts the function of mitochondria and muscles by turning on AMPK, which raises the levels of SIRT1 and PGC-1α and increases the activity of citrate synthase. This helps keep glucose levels stable [112, 186, 187]. Since obesity affects how blood vessels work, RSV may help restore endothelial function. For example, it greatly increased Flow-Mediated Dilation (FMD) in the brachial artery, especially in people who had low FMD responses to begin with [188, 189]. This is in line with previous research showing a dose-dependent association between RSV and FMD improvement, likely due to nitric oxide release via eNOS-3 activation [115, 190, 191].
In a separate study, RSV supplementation in obese individuals reduced postprandial glucagon levels while not affecting fasting levels, potentially averting hyperglucagonemia in individuals with IGT and T2DM [116].
Elevated levels of apolipoproteins, such as ApoB-100 and ApoB-48, are linked to insulin resistance and are commonly seen in obesity, T2DM, CVD, and Chronic Kidney Disease (CKD) [192-194]. A two-week RSV intervention (1000 mg/day in week one and 2000 mg/day in week two) led to a 22% and 27% reduction in ApoB-100 and ApoB-48, respectively, although improvements in fasting insulin and insulin resistance were not statistically significant [117]. These outcomes resemble effects seen with calorie restriction results are similar to those observed with calorie restriction [195–197].
When given to obese men at a dose of 150 mg/day, RSV has also been shown to shrink adipocytes, which is a sign of better insulin sensitivity. This decrease was linked to the activation of autophagy pathways through transcription factor EB and the inhibition of Wnt and Notch signalling, which are pathways pertinent to chronic disease management [198, 199]. A meta-analysis of 11 placebo-controlled trials corroborated RSV's effectiveness in enhancing insulin sensitivity in individuals with diabetes [156]. Synthetic RSV derivatives have also demonstrated encouraging outcomes. For instance, RSV triphosphate showed stronger antioxidant effects and greater bioavailability than native RSV, thereby greatly reducing oxidative stress in obese individuals [119, 200].
RSV also works better when used with other treatments. In obese individuals, the concurrent administration of orlistat resulted in significantly greater weight loss compared to the use of either agent in isolation [120]. Additionally, a co-formulation of trans-RSV and hesperetin elevated glyoxalase-1 expression, diminished plasma methylglyoxal levels, and enhanced glycemic control and insulin sensitivity, indicating potential efficacy in the management of diabetic kidney disease [201-204].
RSV by itself has also been helpful. In patients with Metabolic Syndrome (MS), taking high doses (1500 mg/day for 3 months) caused a lot of weight loss and lower BMI, waist circumference, and insulin secretion [162]. Even though these doses were beneficial, both high (1000 mg) and low (150 mg) RSV doses did not change hsCRP levels or the expression of inflammatory genes. They only had a small effect on glucose and lipid profiles [205]. In a separate study, RSV decreased androgen precursors, such as androstenedione and DHEA-S, but did not affect prostate-specific parameters, suggesting limited applicability in benign prostatic hyperplasia [206]. However, very low doses of RSV (75 mg/day) did not enhance the resting metabolic rate in postmenopausal women, nor did they significantly influence the expression of critical molecular targets such as SIRT1, AMPK, PGC-1α, or UCP3 in skeletal muscle or adipose tissue [182].
RSV shows promise as both an adjunct and a standalone treatment for diabetes, obesity, and metabolic syndrome. However, its clinical efficacy seems to be affected by variables such as age, sex, ethnicity, genetic predisposition, dosing schedule, and concurrent medical conditions. While numerous trials affirm its efficacy, definitive dosing strategies for prolonged use and combination therapy have yet to be established through additional research.
3.10. Pharmacokinetics of Resveratrol and its Analogues
RSV is rapidly absorbed and metabolized, so it is quickly eliminated from the body, contributing to its decreased bioavailability [207]. Moreover, RSV bioavailability varies significantly among individuals and seems to be gender-neutral [208]. By altering how RSV is absorbed and processed, the gut microbiota has a substantial impact on the production of RSV metabolites, which may help to explain some of this variability. It is found that the micronized formulation of RSV has yielded a peak plasma concentration of 1942 ng/mL, which is considered to be much higher than the Cmax of the standard formulation, i.e., 538.8 ng/mL Furthermore, even at a high single dose of 5 grams, SRT501 showed improved bioavailability in cancer patients with no significant side effects [124].
According to pharmacokinetic analyses, higher doses of RSV take roughly 1.5 to 2 hours to reach maximum concentration, whereas lower doses reach peak plasma levels in 30 minutes [209, 210]. RSV absorption may be retarded when taken with food, extending the time it takes for healthy people to reach Cmax [211]. RSV has a wide distribution due to its lipophilic properties, which enable it to accumulate in a variety of organs and tissues, including the liver, intestines, and brain. RSV concentrations varied between 46 and 914 ng/g in normal tissues and 52 to 2834 ng/g in tumor tissues after 14 days of SRT501 administration (5 g/day) [124]. Human plasma, urine, and tissues have been shown to contain approximately 20 RSV-derived metabolites [212, 213]. RSV-3-O-sulfate is the main circulating metabolite in humans, as opposed to RSV-3-O-glucuronide, which is predominant in animal models (such as rats and pigs) [214]. RSV-3-O-glucuronide, 4′-O-glucuronide, and RSV sulfate-glucuronide are additional noteworthy metabolites. At lower doses (≤50 mg), glucuronides are more prevalent [126], while sulfate-glucuronide takes over as the main metabolite after a few hours [113]. Even within the same tissue or organ, metabolite concentrations can vary depending on the biopsy site.
Urine and feces are the main routes of elimination for RSV and its metabolites, though the rate of elimination can vary depending on the compound's formulation or delivery system [215, 216]. Particularly at higher doses, RSV may interact with cytochrome P450 (CYP) enzymes, potentially preventing the metabolism of other medications and increasing systemic exposure to coadministered CYP substrates [217]. Some interactions, however, might be advantageous. For instance, RSV can be converted to piceatannol, a substance with potent anticancer properties, by CYP1B1, which is frequently expressed in tumors [218]. By activating RSV into a more potent form, this enzymatic conversion implies that CYP1B1 may be essential in mediating the virus's cancer-preventive effects [219, 220].
Furthermore, RSV pharmacokinetics have been found to vary with the time of day, with morning doses exhibiting a higher bioavailability [221]. Overall, RSV's absorption, metabolism, and distribution within the body are strongly influenced by factors such as dosage, timing, formulation, and individual physiology.
RSV's therapeutic potential has long been recognized, but several pharmacokinetic issues have prevented its widespread use in healthcare settings. RSV is highly chemically unstable and is very susceptible to photolysis and oxidative degradation, leading to rapid loss of activity. Furthermore, it is quickly eliminated from the body due to its extensive first-pass metabolism, low oral bioavailability, and poor aqueous solubility [222–225]. Formulations based on nanotechnology have been investigated extensively to overcome these constraints. It has been demonstrated that encapsulating RSV in nanoparticles can prevent environmental degradation, enhance absorption, increase bioavailability, and provide controlled or prolonged drug release [226-231]. Its physicochemical stability and biological performance can also be enhanced by methods like complexation with hydroxypropyl-β-cyclodextrin or β-lactoglobulin, as well as encapsulation in liposomes, calcium-alginate submicron particles, zein and gliadin nanoparticles, nanoemulsions, and solid lipid nanoparticles. Together, these cutting-edge techniques help overcome RSV's inherent pharmacokinetic limitations, expanding its use as a therapeutic and nutraceutical agent [232-236].
3.11. Limitations and Challenges with Resveratrol
RSV has demonstrated significant potential in the treatment of human diseases. Yet, few studies have reported undesirable side effects associated with consuming this polyphenol. Researchers have looked into the safety of RSV in both healthy people and people with different health problems. In a preliminary clinical trial, healthy participants were administered various amounts of RSV (25, 50, 100, and 150 mg) at four-hour intervals over 48 hours. A few people said they had mild side effects like headaches, dizziness, and epididymitis [221]. Another study examined the safety of RSV at higher doses (0.5, 1.0, 2.5, and 5.0 g) and found that only the 2.5 and 5.0 grams doses caused mild side effects like nausea, diarrhea, and stomach pain [215]. In a phase I randomized clinical study involving 40 healthy volunteers, a daily dose of 0.5-5 grams of RSV administered over 4 weeks was generally deemed safe, although higher doses elicited mild gastrointestinal symptoms. Major plasma metabolites (sulfates and glucuronides) attained concentrations up to 20-fold greater than the parent RSV. The findings showed that RSV lowered circulating IGF-1 and increased IGFBP-3 levels with the strongest effect at 2.5g/day. This suggests that RSV may help protect against cancer [148].
Other studies have also found that taking RSV can cause diarrhea, headaches, and hot flashes [211, 238]. Patients with liver metastases who took RSV reported feeling sick [124]. When given to people with Type 2 Diabetes Mellitus (T2DM) at a dose of 3 mg/day for 12 months, RSV caused different side effects, such as a mild increase in ALT, low blood sugar, and diarrhea in some participants [237, 238]. Additionally, RSV has antiplatelet effects, which could raise the risk of bleeding and bruising, especially in people who are also taking anticoagulant or antiplatelet drugs like clopidogrel, enoxaparin, dalteparin, heparin, warfarin, or NSAIDs. Because these drugs already thin the blood, taking them with RSV could make bleeding problems more likely [239-241].
In general, RSV has been associated with certain adverse consequences, but they are usually mild and don't occur very often, especially when weighed against the numerous advantages it provides for improving health.
4. DISCUSSION AND FUTURE PERSPECTIVES
Many preclinical and animal studies exhibit that RSV has numerous health benefits. Nevertheless, due to differences in bioavailability, metabolism, and dosage methods, such findings often fail to lead to significant conclusions in clinical trials involving humans. The capacity of the body to absorb and use RSV is severely hampered by its inability to dissolve in water, high metabolism, and chemically unstable nature. These are among the reasons for its reduced bioavailability. There are several ways to make it more useful as a medicine and more readily available to the body. Micronization can reduce the size of RSV particles, increasing the surface area available for absorption. Adding excipients may improve absorption by increasing drug solubility and prolonging its time in the gastrointestinal tract.
To ensure customer safety and maintain the same high quality of these RSV formulations across global markets, there must be strict rules for verifying their purity using advanced analytical methods.
To determine the therapeutic benefits, optimal dosages, and mechanisms of these formulations in addressing conditions such as diabetes, cancer, obesity, dyslipidaemias, and inflammatory diseases, meticulously designed long-term clinical trials should be undertaken subsequently. If more research is conducted on RSV and its derivatives, they could be used as adjuvants for neurodegenerative diseases.
CONCLUSION
Numerous RSV analogues have been identified in recent years, demonstrating significant therapeutic potential owing to their enhanced potency, improved pharmacokinetics, and multitargeted activity. These molecules suggest they could aid in the management of conditions such as cancer, inflammation, neurological problems, and metabolic issues like diabetes and obesity. Several patents highlight their potential importance in Alzheimer's disease, a condition with limited therapeutic alternatives. Even though there is promising preclinical evidence and early clinical trials, RSV's therapeutic use is limited by problems like low bioavailability and fast metabolism. However, it is considered safe at doses up to 5 g/day and can modify numerous disease markers when given alone or in combination with other phytochemicals. RSV works differently from regular single-target drugs because it can control multiple signalling pathways. This makes it useful for managing chronic diseases. Its long history of being used as a food, its safety record, its low cost, and its ease of access all make it a popular medicine. RSV has not yet received approval for the treatment of any specific human disease; however, ongoing research into its structure-activity relationships and molecular mechanisms may facilitate broader clinical application. RSV shows great promise as a natural, multi-targeted treatment option for both preventing and treating a range of human diseases, yet more research will be required to confirm this.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: M.K.: Conceived the idea and coordinated the overall writing of making manuscripts, including the use of PRISMA; N.P.: Wrote the original draft and conducted the detailed survey for patents and clinical trials; R.G.: Reviewed and edited the manuscript; K.N.: Helped with the overall visualization of the manuscript. All authors read and approved the final manuscript.
LIST OF ABBREVIATIONS
| HbA1c | = Glycosylated haemoglobin |
| IL | = Interleukin |
| ALP | = Alkaline phosphatase |
| ALT | = Alanine aminotransferase |
| CCL3 | = Chemokine (C‐C motif) ligand 3 |
| TNF‐α | = Tumor necrosis factor α |
| AMPK | = 5′‐AMP‐activated protein kinase |
| PGC‐1α | = Peroxisome proliferator‐activated receptor‐γ coactivator 1α |
| LGR5 | = Leucine‐rich repeat‐containing G‐protein‐coupled receptor 5 |
| PSADT | = Prostate‐specific antigen doubling time |
| RASSF | = Ras association domain family |
| NAFLD | = Nonalcoholic fatty liver disease |
| NF‐κB | = Nuclear factor‐κB |
| NIHSS | = National Institutes of Health Stroke Scale |
| COX‐2 | = Cyclooxygenase 2 |
| IKKβ | = Inhibitor of nuclear factor κB kinase subunit β |
| CRP | = C‐reactive protein |
| JNK | = C‐Jun N‐terminal kinase |
| LDL | = Low‐density lipoprotein |
| VCAM‐1 | = Vascular cell adhesion molecule 1 |
| VEGF | = Vascular endothelial growth factor |
| PCOS | = Polycystic ovary syndrome |
| Ang‐2 | = Angiopoietin 2 |
| Tie‐2 | = TEK receptor tyrosine kinase |
| T2DM | = Type 2 diabetes mellitus |
| TSP‐1 | = Thrombospondin 1 |
| ICAM‐1 | = Intercellular adhesion molecule 1 |
| hsCRP | = High‐sensitivity C‐reactive protein |
| PAI‐1 | = Plasminogen activator inhibitor type 1 |
| Flk‐1 | = Fetal liver kinase 1 |
| AUH | = Aarhus University Hospital |
| CKD | = Chronic kidney disease |
| CRC | = Colorectal cancer |
| CVD | = Cardiovascular disease |
| DTCGH | = Dalin Tzu Chi General Hospital |
| ED | = Endothelial dysfunction |
| IGT | = Impaired glucose tolerance |
| KTPH | = Khoo Teck Puat Hospital |
| MUMC | = Maastricht University Medical Center |
| NRC | = National Research Council |
| SUMS | = Shiraz University of Medical Sciences |
| UMCC | = University of Michigan Cancer Center |
| UMN | = University of Michigan |
AVAILABILITY OF DATA AND MATERIALS
All the data and supportive information are provided within the article.
CONFLICT OF INTEREST
The author, Dr. Monika Kaurav, is the Editorial Advisory Board member of The Open Biotechnology Journal.
ACKNOWLEDGEMENTS
The authors would like to express their gratitude to Dr. Adesh Kumar Pandey, Director of Academics, and Dr. Manoj Goel, Executive Director of the KIET Group of Institutions, India, for their support and motivation.


