All published articles of this journal are available on ScienceDirect.
New Perspectives in the Treatment of Tumor Cells by Electromagnetic Radiation at Resonance Frequencies in Cellular Membrane Channels
Abstract
Background:
The use of electromagnetic fields has been considered as adjuvant therapy for the treatment of cancer given that some clinical trials have shown that the irradiation of cancer cells with electromagnetic fields can slow down the disease progression.
Aims:
We hypothesize that this effect could be amplified by irradiating tumor cells with electromagnetic fields having frequencies close to the natural resonant frequencies of membrane channels in tumor cells, in order to obtain a significant change of the ion flux across tumor cell membrane channels, inducing the largest harmful alteration in their cellular function.
Methods:
Neuronal-like cells were used as a cell model and exposed for 6 h to electromagnetic fields at different frequencies (0, 50 Hz, 900 MHz) at the same intensity of 2 mT. The exposure system was represented by two Helmholtz coils driven by a power amplifier in current mode and an arbitrary function generator. FTIR spectroscopy was used to evaluate the results of the exposure.
Results:
The results of this study showed that the Amide I vibration band increased in intensity with the increase of the frequency, leading us to assume that the displacement of the cell channels α-helices depends on the frequency of the applied electromagnetic fields.
Conclusion:
This preliminary result leads us to plan future research aimed at searching for the natural frequencies of membrane channels in tumor cells using resonant electromagnetic fields in order to damage the cellular functions of tumor cells. Clinical trials are needed to confirm such a hypothesis derived from this physical study.
1. INTRODUCTION
One of the most serious diseases that has developed in the modern era is certainly cancer.
At this moment the methods of cancer treatment usually used are surgery, radiosurgery, radiotherapy, chemotherapy and immunotherapy. Unfortunately, the use of these techniques is not always effective mainly due to side effects, because even normal cells are affected by such therapies, inducing the oncologists to limit the same therapy that was chosen [1-7].
In recent years, the use of electromagnetic fields has also been considered as adjuvant therapy for the treatment of cancer. In this regard, some results have shown that the irradiation of cancer cells with electromagnetic fields has slowed the disease progression without inducing significant side effects [8-14].
The harmful effects of electromagnetic fields on cancer cells can be easily explained by the results of previous studies showing that protein unfolding and aggregation occur under exposure to electromagnetic fields [15-22] so that the irradiation of cancerous cells with an electromagnetic field should alter their cellular functions, causing them to die.
In order to amplify this effect, it is plausible to think of irradiating tumor cells with an electromagnetic field having frequencies equal to (or at least close to) the natural frequencies of the tumor cells that are to be treated, in order to obtain the largest alteration in their cellular function.
This hypothesis derives from the resonance theory, according to which, displacements of molecules are amplified at natural resonant frequencies of the same material.
However, the problem is to search for the natural resonance frequencies of tumor cells. For this aim, some mechanical models have been provided up to now. For instance, natural resonant frequencies of cells were assumed to depend on some mechanical properties (the dimension of the cell, density and elastic modulus), so that the natural resonant frequencies of normal cells are different from the natural resonant frequencies of cancer cells [13]. Particularly, a model was proposed in recent studies, derived from civil engineering principles, for computing the natural resonance frequency of cancer cells in order that an applied external electromagnetic field can interact with the cancer cell at that frequency inducing a resonance, damaging the cancer cells [23].
Another study proposed that measuring Young’s modulus and cell shape as a function of the frequency of a mechanical vibration can be used for computing the natural frequency of spreading osteoblast cells [24].
Nevertheless, the models proposed in the studies mentioned above are based on the mechanical properties of cells, so that the possible resonant interactions strictly depend on mechanical properties that cannot be considered as the most important characteristics related to cellular functions. In contrast, a physical-chemical parameter which has an important role in cell viability is represented by ion flux in cellular membrane channels. Indeed, it was shown that ion flux across cell channels has a fundamental role in cellular functions as it can regulate the initiation, progression and proliferation of cancer [25-29].
Hence, resonance interaction mechanisms between an applied electromagnetic field and membrane channels in cancer cells can be hypothesized, in order to significantly alter the ion flux across cell membrane channels, damaging cancerous cells, as described in the following sections.
2. MATERIALS AND METHODS
Protein α-helices are present in all types of cellular membrane channels [30-32]. In particular, cellular membrane protein accounts for about 50% of its mass [33]. Taking advantage of this circumstance, a preliminary study was carried out studying protein response to electromagnetic fields at different frequencies by means of Fourier Transform Infrared (FTIR) spectroscopy.
Neuroblastoma cell line SH-SY5Y was purchased from American Type Culture Collections (ATCC) (Rockville, MD) and was used as a cell model in this study. Cells were differentiated following the protocol accurately described in the previous studies [16, 17].
The exposure system was represented by two Helmholtz coils, used to produce a uniform magnetic field at the center of the distance between the coils, following Helmholtz coils theory. The Helmholtz coils were driven by a 7570 AE power amplifier in current mode (Techron, Elkhart, IN) and a Model 75 arbitrary function generator (Wavetek, Plainview, NY), that were used to generate a Direct Current (DC) and an Alternating Current (AC) at 50 Hz frequency. Moreover, microwave sources at frequencies ranging up to 900 MHz were obtained using mobile phone devices. Further technical characteristics of the experimental setup used in this study have already been described [34, 35].
However, contrary to the protocol used in previous studies by the authors, different cell samples were exposed for 6 h to electromagnetic fields at different frequencies (0, 50 Hz, 900 MHz), but the same intensity of 2 mT was used for the three types of electromagnetic fields. This protocol was chosen in order to compare the effects of electromagnetic fields at different frequencies.
Neuronal-like SH-SY5Y cells, grown in 25 cm2 culture flasks, were located in the center of the field generated between the coils. The coils and cell samples were placed into an incubator (series 5400-115V models, Thermo Electron, Winchester, VA) with 5% CO2/95% humidity and at the temperature of (37.0 + 0.1) °C. Unexposed samples were located in another incubator, at the same physical conditions reported above.
Cell cultures were treated following the protocol of [35, 36] and subjected to infrared (IR) spectroscopic techniques by a FTIR spectrometer Vertex 80v of Bruker Optics. For each spectrum, 128 interferograms were collected with a spectral resolution of 4 cm-1, following the technical procedure described previously [16, 17].
3. RESULTS
Representative FTIR spectra of neuronal-like cells samples after 6 h exposure to static, 50 Hz and 900 MHz electromagnetic fields at 2 mT are reported in Fig. ( 1A- 1C), respectively. The most evident vibration band appearing in these spectra is represented by an intense Amide I band centered around 1650 cm-1, corresponding to the protein α-helix structure, that is due to C=O stretching and N―H bending vibrations and a weak Amide II vibration centered at 1545 cm-1, that is due to the N―H bending and C―N stretching vibrations.
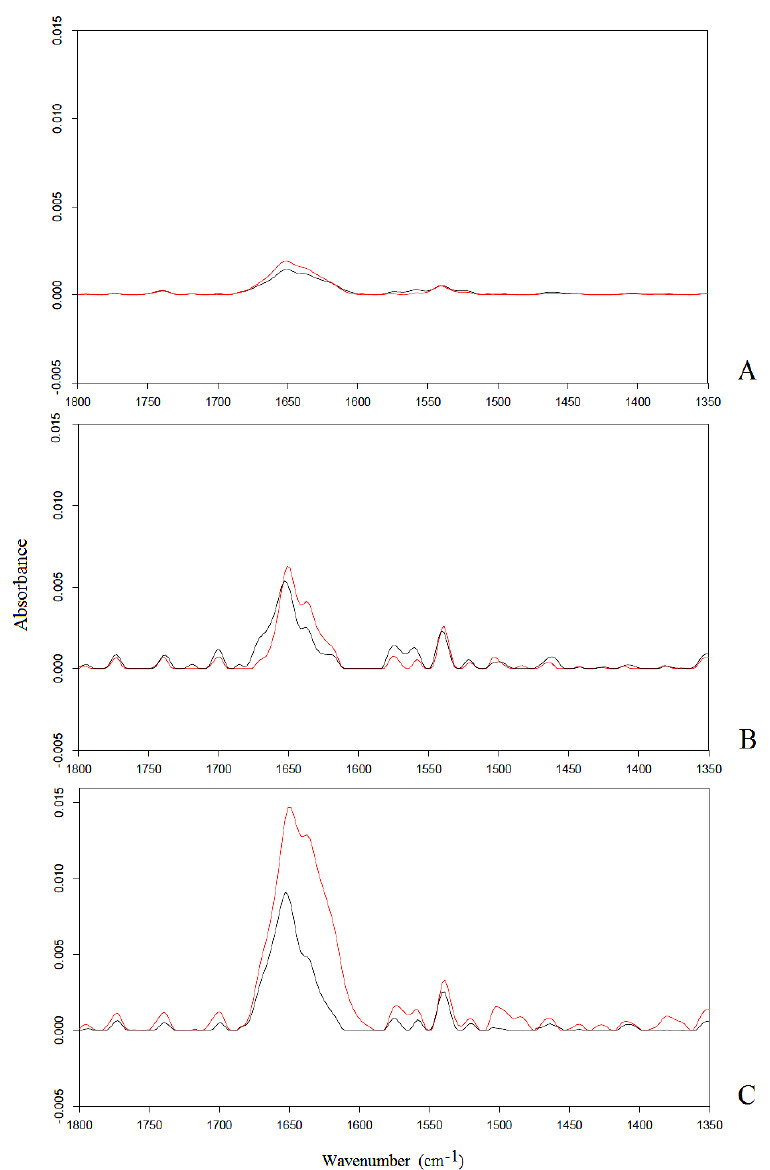
A significant increase in the intensity of the Amide I band was observed after exposures to 2 mT electromagnetic fields at 0, 50 Hz, 900 MHz, with p < 0.05 significance. This result was already explained assuming that protein α-helices had aligned with the applied electromagnetic fields, showing that the exposure of proteins in bidistilled water solution to an applied electromagnetic field causes a torque which induces the alignment of protein α-helices towards the direction of the applied field [34,37-40]. As a result, an enlargement in the diameter of cellular membrane channel should occur, inducing a decrease in the resistance of the channel and a consequent increase of ion flux, changing the delicate equilibrium of cellular functions [34].
Interestingly, the integrated area of the Amide I band increased significantly (p < 0.05) with the increase of the frequency of the applied electromagnetic field, demonstrating that the displacement of protein α-helices is closely dependent on the frequency of the applied electromagnetic field.
4. DISCUSSION
The results of this study have induced us to assume that tumor cells can be irradiated by an electromagnetic field at a frequency that is very close to the natural resonant frequencies of cancer membrane channels, producing a resonance phenomenon which should induce the maximum displacement of channels α-helices, amplifying ion flux across the same channels following the 2nd law of Ohm, as schematized in a figure [34].
Previous studies on the effects of radiation therapy showed that the use of beams of X-rays, gamma rays or particle radiation (protons or heavy ions) towards targeted areas can also damage normal non-irradiated cells neighboring tumor cells, a phenomenon termed ‘bystander effect’ [41-45]. This effect still represents an important limitation to the use of radiation therapy for the treatment of cancer.
In contrast, by following resonance theory, neighboring normal cells would not be damaged significantly by the irradiation of an electromagnetic field at the natural resonant frequency of cancer cell membrane channels, because the natural resonant frequencies of normal cells should be significantly different from the natural resonant frequencies of tumor cells.
Furthermore, it has been shown that even a minimum alteration of ion flux across cell membrane channels induces relevant cell damage [46, 47], so that the resonant mechanism described above can be successfully used for cancer treatment.
The angular deviation of α-helices in membrane channels of tumor cells can be measured as a function of the frequency of the electromagnetic field to which the tumor tissue is exposed. The maximum displacement of channels α-helices should occur at the natural resonance frequency of the cancerous cells which are being analyzed, inducing their death. To this aim, spectroscopic circular dichroism can be used to measure the angular deviations of α-helices in membrane channels of tumor cells as a function of the frequency of an applied electromagnetic field, using variable frequency power generators.
In addition, patch clamp technique can be used for measuring ionic currents across cellular membrane channels of the tumor tissue as a function of the frequency of an applied electromagnetic field, so that the largest value of ionic current would be measured at the natural resonance frequency of tumor cells. Furthermore, the largest decrease in cellular transmembrane potential in a tumor cell should occur at the natural resonant frequency, following the results obtained in previous studies by the authors of this study [16, 17].
Analogue measurement of the physical-chemical parameters cited above can be carried out in neighboring “non-resonant” normal cells, in order to verify that significantly lower values are obtained in normal cells in comparison with those obtained in cancer cells, in order to confirm the absence of a bystander effect after the irradiation by resonant electromagnetic fields of the targeted areas.
Once the natural resonant frequencies of cellular channels α-helices in typical cancerous tissues have been found, experimental verification of results would be carried out by measuring the disease progression in living beings after irradiation by electromagnetic fields at resonant frequencies.
CONCLUSION
Given that cancerous tissues should have natural frequencies quite different from the natural frequencies of healthy tissues, it can be hypothesized that irradiating tumor tissues by electromagnetic fields at frequencies close to the natural frequencies of tumor cells membrane channels would cause resonant interaction with membrane channels α-helices, thereby altering ion flux across them and damaging cellular functions of cancer cells. However, such a perspective should be confirmed by biochemical analyses and medical approaches.
Such resonant frequencies can be obtained for each tumor tissue by both spectroscopic and biochemical measurements of channels α-helices rotation and ion flux across cell channels as a function of the frequency of electromagnetic fields. In any case, clinical trials are needed to evaluate the possible application of such a result.
DISCLAIMERS
The views expressed in the submitted article are our own and not an official position of the institution or funder. The corresponding author is responsible for statistical analyses on the title page. No financial support was received to carry out this study.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIAL
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We are grateful to Prof. Riccardo Ientile (Department of Biomedical Sciences and Morpho-functional images, University of Messina, Italy) for his kind and precious collaboration regarding the preparation of SH-SY5Y neuronal-like cells samples. The corresponding author is responsible for statistical analyses on the title page.


