All published articles of this journal are available on ScienceDirect.
A Review of Three-dimensional Printing for Biomedical and Tissue Engineering Applications
Abstract
Background:
Various living organisms especially endangered species are affected due to the damaged body parts or organs. For organ replacement, finding the customized organs within the time by satisfying biomedical needs is the risk factor in the medicinal field.
Methods:
The production of living parts based on the highly sensitive biomedical demands can be done by the integration of technical knowledge of Chemistry, Biology and Engineering. The integration of highly porous Biomedical CAD design and 3D bioprinting technique by maintaining the suitable environment for living cells can be especially done through well-known techniques: Stereolithography, Fused Deposition Modeling, Selective Laser Sintering and Inkjet printing are majorly discussed to get final products.
Results:
Among the various techniques, Biomedical CAD design and 3D printing techniques provide highly precise and interconnected 3D structure based on patient customized needs in a short period of time with less consumption of work.
Conclusion:
In this review, biomedical development on complex design and highly interconnected production of 3D biomaterials through suitable printing technique are clearly reported.
1. INTRODUCTION
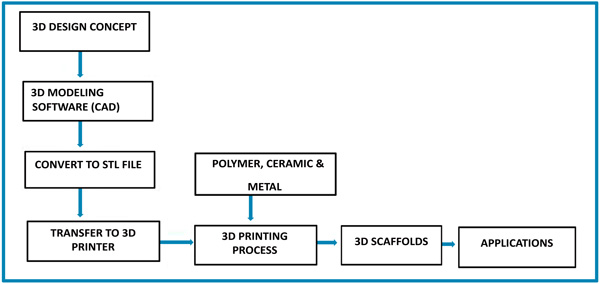
Three-dimensional printing was first patented in 1986 by Charles Hull for Stereolithography Apparatus (SLA). Early researchers are known as Rapid prototyping technologies. Later, stereolithography is commonly known as 3D printing. 3D printing was initially used to create prototypes for product development within certain industries. Dr. Hideo, a Japanese lawyer was the first person to file a patent for rapid prototyping technology. Charles (chuck) Hull was the first person to invent the stereolithography machine (3D printer), which was the first ever device of its kind to print a real physical part from a digital (computer) generated file [1, 2]. Three-dimensional printing technology is one of the trending additive manufacturing methods. It is a process of making a 3D object by adding layer-by-layer of required material using a three-dimensional digital model [3, 4]. The most commonly used core material for additive manufacturing includes ceramic, metal, plastic and polymers (synthetic or natural polymers) [5-7]. 3D objects are mainly formed under the efficient control of digital computer, 3D modeling software (computer aided design or computer tomography scan images), machine equipment and layering materials [8]. Stereolithography is one of the commonly used software file types that is used for 3D printing [9]. After the given CAD model, 3D printing reads the input data from 3D modeling software. Finally, the highly sophisticated 3D objects are manufactured, which can easily produce tedious shapes and structures [10]. The three-dimensional printing process is shown step-by-step in Scheme 1.
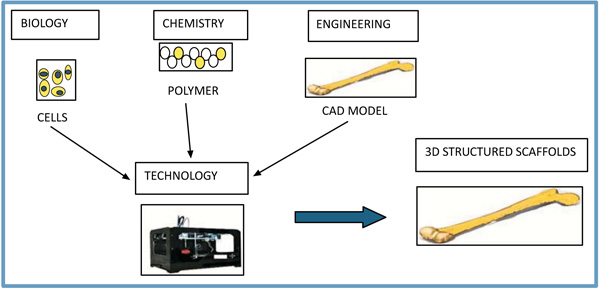
Typically, many additive manufacturing processes are available in industries, laboratories, etc. It majorly integrated several fine parts such as vat photopolymerization, material extrusion, powder bed fusion, binder jetting, as shown in Table 1. Stereo-lithography (SLA) comes under the basics of vat photo-polymerization method [11, 12]. It is widely recognized as a first 3D printing method. SLA is a laser-based process, which mainly works with photopolymer resins to form a solid 3D object. In this process, photopolymer resin is finely placed in a VAT with a movable platform inside. A laser beam is sharply focused on the surface of the resin, and 3D structures are formed using CAD. Stereo-lithography is one of the most significant 3D printing processes with the good surface finish. Digital light processing is also depending on VAT photopolymerization. The huge difference between them is the light source, and it produces highly accurate parts with excellent resolution. The most widely used method in the material extrusion process is the Fused Deposition Modeling (FDM) or Fused Filament Fabrication (FFF) method. Fused Deposition Modeling uses continuous thermoplastic filament as the printing material to form fine structures and scaffolds [13-15]. In this process, it works by melting plastic filament in nozzle head and precisely deposit in build platform to form 3D structure according to the 3D data supplied to the printer. FDM process needs support structures for various bio-applications with overhanging geometries. FDM process is a highly accurate and reliable process that is studio-friendly. In the powder bed fusion process, the most widely used method is the Selective Laser Sintering (SLS) technique for scaffold fabrication. SLS is an additive manufacturing technique, which uses high power laser as a power source to sinter powdered material to fabricate various 3D scaffolds by 3D model [16, 17]. The significant material used in SLS are plastic, metal, ceramic, glass powders. SLS has the potential for creating prototypes, scaffolds, models and even final products, and it is mostly utilized in industry and all medical fields. It provides highly complex parts with adequate interior components and is the fastest Additive Manufacturing (AM) process for printing three-dimensional functional parts and organs with designed structural integrity. In Inkjet printing, the material being jetted is a binder, and it selectively drops into powdered bed of the part material to fuse for creating three-dimensional objects and scaffolds for various medical applications. A range of different materials can be used for three-dimensional printing for various fabrications [18, 19]. Different areas of the scientific community combined to form a 3D structure, which is shown in Scheme 2.
| Methods | Classifications | Ref. |
|---|---|---|
| Vat photopolymerization | i) Stereolithography (SLA) ii) Digital Light Processing (DLP) |
80-84 |
| Material extrusion | i) Fused Deposition Modeling (FDM) | 97-99 |
| Powder bed fusion | i) Selective Laser Sintering | 112-114 |
| Binder jetting | i) Inkjet 3D printing | 122 |
3D file of the object can be created using Biomedical CAD software, with a 3D scanner. The variety of materials can be used for printing purposes such as plastics, alumide, ceramics, resins, metals, sand, textiles, biomaterials, glass, food and even lunar dust. Fused Deposition Modeling and Selective Laser Sintering use plastic and alumide for fabricating 3D scaffolds. Some FDM printers have two or more print heads to print multiple variable colors in scaffolds. Selective Laser Sintering consists of fabrication of 3D object by melting successive layers of powder together to form a scaffold. Stereolithography and Digital Light Processing use photopolymerization for developing highly sophisticated 3D products [20, 21]. 3D printing is excellent for developing healthcare products in many ways, including implantable and non-implantable medical devices along with cost-effective customizable devices, patient-specific products in orthopedics and maxillofacial surgery, fabricating human living tissue, prosthetics and accurate pre-op models for academic purpose. It is mainly helpful for fabricating different types of living tissues, recreating difficult bone disorders such as craniofacial disfigurement, hearing aids and dental delivery devices, which offer excellent visualization and great dimensional stability. 3D printing also creates medical fixtures, functional testing models, industrial design and end-use parts [22, 23].
Nowadays, the ultimate aim of tissue and organ engineering is to restore normal functions of living organs and tissues, regeneration, replacement of defective or injured organs and tissues using different technology. To achieve this aim amongst different technology, three-dimensional scaffolds are commonly used for biomedical, tissue engineering applications and all medical fields, which is made up of polymer (natural or synthetic polymers), metals, ceramics, etc. Three-dimensional Scaffolds provide several mandatory functions [24-26]. It creates an adequate internal pathway for the cell attachment and migration. It should transfer several growth factors, oxygen transport and waste product removal in 3D scaffolds [27, 28]. It must be biocompatible, good mechanical properties and keeps its shape and structure when the cells and tissue are growing [29-32]. Hence, 3D bioprinting technology is commonly used for controlling cell and tissue pattern to be retained viability and functionality of the cells inside the printed 3D structure using different biomaterials [33-35]. Many researchers have been studied development and improvement of the appropriate scaffold using 3D printing in tissue engineering applications. Advances developed by 3D printing enhance the ability to control pore volume, pore size, and pore interconnectivity in 3D scaffolds for patient specific applications. In addition, materials used in a 3D printing machine, which is very essential for fabricating 3D scaffolds using 3D modeling software and scan data. 3D bioprinting process uses living cells and bioactive molecules in biomaterials, which produces a 3D structure that does not affect the viability and functionality of the cells [36, 37].
In future, researchers should be considered in the biomaterials such as bio-ink for creating 3D objects in tissue engineering applications and all the medical fields. Recent advancement in the biomedical field of stem cell development can be approached to the Bio3D printing cells fabrication techniques. It has the huge potential of studying disease modelling, discover drugs and mimicry of cellular components. The microfluidic approach in 3D tissue fabrication printing has garnered to a significant leap in the vascularization of biomedical engineering.3D printing technology can be evolved to cover the entire range of biomedical applications beginning from diagnosis and ending with prognosis. The potential of 3D printers can be exploited in the field of biomedical engineering such as research works, drug delivery, lab testing, clinical practice and helping the surgeons with as detailed mock surgeries as possible. 3D printing is used as a one-step solution for all the biomedical engineering problems [38-40].
Among different rapid prototyping technologies, the widely used four technologies for biomedical applications are stereolithography, fused deposition modeling, selective laser sintering and inkjet 3D printing [41-43]. Even though these techniques are used in various fields such as architectural modeling, art, lightweight machines, aircraft industry, defense field and medical fields, but it is excellent in tissue and other biomedical engineering applications [44-48]. Many researchers keep on improving various methods and materials to create 3D structure by satisfying the mechanical properties, biocompatibility for regeneration of normal tissues and bone regeneration, etc. [49, 50]. In this review, only few ongoing technologies are discussed, which can produce highly precise, greatly customized and extremely interconnected bio parts by satisfying all requirement of biomedical needs.
2. THREE-DIMENSIONAL PRINTING FOR TISSUE ENGINEERING APPLICATION
Three-dimensional printing technologies are an emerging technology to develop new tissues and organs [51-55]. Many researchers are currently conducting a study for fabricating 3D structure, which is useful for tissue engineering fields [56-60]. Three-dimensional bioprinting creates unique 3D structure, which controls cell proliferation, attachment and migration within 3D printed structures [61-65]. Therefore, different types of three-dimensional bioprinting techniques are used for a variety of tissue and organ engineering applications [66-70]. Herein, we will discuss the four different types of three-dimensional bioprinting methods, which are most commonly used methods such as Stereolithography and Digital Light Processing in Vat Photopolymerization, Fused Deposition Modeling in material extrusion, Selective Laser Sintering in powder bed fusion and Inkjet printing in binder jetting methods. Table 2 represents some of the advantages and disadvantages of different 3D printing methods in tissue engineering applications.
| Methods | Advantages | Disadvantages | Materials | Ref. |
|---|---|---|---|---|
| SLA, DLP | Simple and complex Fast and good resolution |
Expensive equipment and materials | PEG, PCL, PEGDA | [79-84] |
| FDM | Easy to use good mechanical properties | Filament required Cannot used with cells |
PCL, PLGA | [96-99] |
| SLS | No need for support materials Various of biomaterials | Rough surface Expensive equipment |
PCL/HA, PCL | [111-114] |
| Inkjet | Cells and hydrogel printed, incorporation of drug and molecules | Low resolution Low mechanical properties |
Fibrin, Gelatin | [121-122] |
2.1. Stereolithography (SLA)
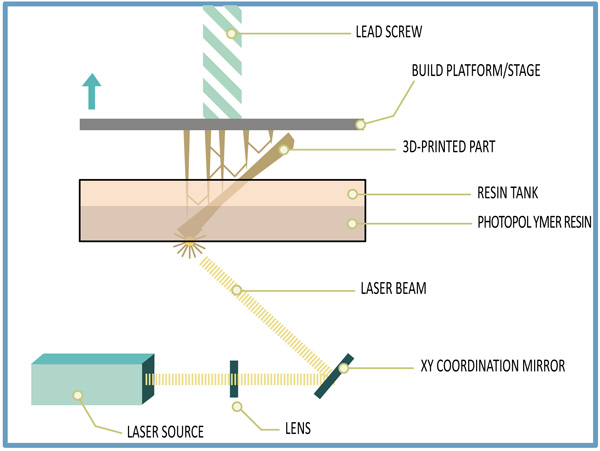
Stereolithography is also known as photo-solidification, which has been early and still widely used 3D printing method. SLA technique has commonly been used to fabricate 3D models, prototypes, patterns and production parts by using UV light in layer-by-layer. This technique has been obtained the patent by Charles (chuck) Hull in 1986 [71-73]. SLA method has the potential to create 3D scaffolds using photopolymerization. Similarly, the DLP technique has also been utilized to create 3D functional models and positive mold objects using visible light source [74, 75]. Both Stereolithography and DLP are formed on the vat photopolymerization [76-78]. In this process, UV light beam is directed onto the area of vat filled with a liquid photopolymer. UV light makes chains of molecules to bind and form polymers. And those polymers are essentially focused to fabricate three-dimensional objects. However, Photopolymerization method is formed free radicals that can affect cell membrane, protein and nucleic acids. Using computer Aided manufacturing or computer Aided design, Stereolithography can make any design that can be fast and expensive [79-81]. A typical schematic of stereolithography technique is shown in Scheme 3. Many scientists obtained the SLA 3D product using various biomaterials in tissue engineering applications. Elomaa et al. fabricated cell-laden hydrogels constructs with biomimetic complexity for use in pharmaceutics, vascular and tissue engineering application. They reported that they used water-soluble methacrylated poly (ethylene glycol-co-depsipeptide) to synthesize and formed a biodegradable photocrosslinkable macromer for SLA [82]. Neiman’s et al. created three-dimensional(3D) hydrogel scaffolds with open channels for post-seeding using photopolymerizable PEG in Stereolithography based method. They showed that structural and functional development of foster formation in 3D liver aggregates. The aim of this study was to develop a platform for drug toxicity study, liver pathophysiology and obtained micro perfusion flow within the open channels of this 3D hydrogel structure [83]. Justinas et al. utilized direct laser writing lithography to fabricate three-dimensional (3D) microstructured scaffolds for cartilage tissue engineering using ultrafast pulsed lasers. They reported that 3D microstructured scaffolds are excellent in spatial resolution, geometry complexity and hexagonal pore shaped hybrid organic and inorganic material micro-structured scaffold, which were fabricated using DLW technique in combining with Cho seeding [84]. Owen et al. created Polymerized High internal Phase Emulsions scaffolds using emulsion templating by combining with micro-stereolithography, which produces cell ingrowth, plasma penetration, tightly controlled and highly interconnected microporosity. Scaffolds constructed using two acrylate monomers with isobornyl acrylate and supported osteogenic differentiation of mesenchymal cells [85]. Du et al. created ceramic artificial bone scaffolds using stereolithography with acrylic resin, which produced correct external shape and internal architecture for bone tissue ingrowth [86]. Hang et al. produced a three-dimensional (3D) scaffold with desired architectures using Stereolithography (SLA) technique. They showed that they used two lentiviral gene constructs with human bone marrow-derived mesenchymal stem cells into a solution of photocrosslinkable gelatin, which was focused using visible light-based projection [87]. Main advantages of stereolithography in tissue engineering applications are fast speed, good resolution, easy to remove support materials, complex designs and fabrication of a simple, and the disadvantages in SLA are a limited range of photosensitive resin and polymers, expensive equipment and materials, cytotoxicity of uncured photoinitiator.
2.2. Fused Deposition Modeling (FDM)
FDM is known as Solid-based AM technology. It is also an Additive Manufacturing technology (AM) which is mainly used for modeling, prototyping and production applications [88-90]. Fused Deposition Modeling was developed and commercialized by Scott Crump and Stratasys, which works under the controlling of Stereolithography (STL) file [91-93]. In this process, FDM printers use a continuous filament of a thermoplastic material in a material extrusion method. These filaments are heated to the melting point temperature, and molten material from the printhead nozzle is deposited on the surface of the growing workpiece to create 3D structures [94]. The nozzle and substrate are controlled by computer to print defined shape and structure, and nozzle can be travelled in both horizontal and vertical directions. Using computer-aided technology, FDM is very flexible to print 3D objects [95-97]. FDM used the thermoplastics PLA, ABS, ABSi, polyphenylsulfone, polycarbonate and among others material. Fused Deposition Modeling is a thermal heating technique, which is used for 3D scaffolds fabrication in tissue engineering applications. Many researchers were investigated using FDM method for tissue engineering applications. Pati et al. fabricated 3D printed scaffolds using a composite of polycaprolactone, polylactic-co-glycolic acid, β- tricalcium phosphate and mineralized ECM laid by human nasal inferior turbinate tissue-derived mesenchymal stromal cells. They studied that they improve the biological functionality of 3D printed synthetic scaffolds to mimics bony microenvironment using Fused Filament Fabrication, and they develop cellular responses and drive osteogenesis of stem cells. Jensen et al. created a polycaprolactone (PCL) scaffold using the combination of Fused Deposition Modeling and thermal induced phase separation to create nanoporous structure in polycaprolactone (PCL) scaffold. They studied PCL3D scaffold to be an excellent osteoconduction and osteointegration [98]. Xu et al. fabricated polycaprolactone nano-HA and polycaprolactone (PCL) 3D artificial bones to mimic natural goat femurs using Computed Tomography-guided Fused Deposition Modeling. They reported that polycaprolactone (PCL) /HA 3D artificial bones scaffolds are excellent in cell biocompatibility, biodegradation ability and bone formation ability, good biomechanical properties that reduce the stress shielding effect [99]. Idaszek et al. created a ternary polycaprolactone (PCL) scaffold, which consists of polycaprolactone (PCL), TCP and PLGA using Fused Deposition Modeling. They evaluated mechanical characteristics, degradation kinetics and surface properties through in-vitro. They resulted that the introduction of PLGA improved the degradation rate and surface roughness [100]. Li et al. used the Fused Deposition Modeling technique to make a comparison between Ti cage and PCL-TCP scaffold as a spinal fusion cage. They reported that inferior fusion performance of the PCL-TCP scaffold at 6 months is similar to Ti cage at 12 months. In addition, PCL-TCP scaffold is resulted in better bone ingrowth and distribution compared to Ti cage [101]. The main advantages of Fused Deposition Modeling are easy operation and easy use, adequate mechanical properties, low cost, solvent is not required and various lay- down patterns and the disadvantages in Fused Deposition Modeling are materials in filament form (thermoplastics), low speed, cannot used with cells and tissues, high temperature and smooth surface.
2.3. Selective Laser Sintering (SLS)
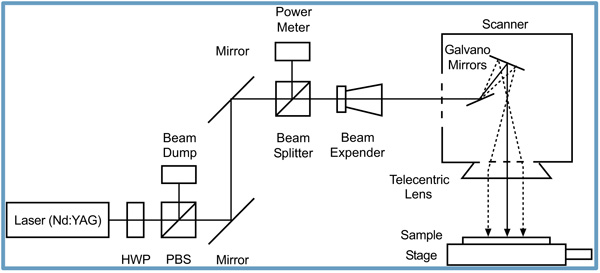
Selective Laser Sintering is an additive manufacturing technique, which uses laser as a power source to form solid 3D scaffolds [102-104]. It is very similar to Direct Metal Laser Sintering. SLS was patented and developed in mid1980’s by Carl Deckard and Joe Beaman. SLS uses high powered lasers, which is too expensive. In this process, high powered lasers selectively fuse powdered material using CAD file or Scan data to form 3D objects [105]. This process is printed various materials; plastic, metal, ceramic and polymers and their composites [106-108]. Moreover, the SLS technique does not require a separate feeder for support material. SLS technique is the ability to make highly complex geometry directly from digital CAD data [109-111]. A typical schematic of Selective Laser Sintering is shown in Scheme 4. Many researchers reported the SLS product using various biomaterials. In addition, SLS is used in tissue engineering applications as scaffolds from various biomaterials and their composites. Du et al. constructed three-dimensional bone scaffolds in Selective Laser Sintering technique with uniform multi-scaled porosity, moderate mechanical properties and good biocompatibility using PCL microspheres, and polycaprolactone /hydroxyapatite composite microspheres are used as the basic building materials. They showed that SLS derived scaffolds are excellent in multiple stem cells behavior, promoting cell adhesion, supporting cell proliferation, inducing cell differentiation, histocompatibility and adequate mechanical features [112]. Chen et al. fabricated polycaprolactone scaffolds for cartilage tissue engineering in craniofacial reconstruction using Selective Laser Sintering technique, which was surface modified through immersion coating with either gelatin or collagen. They reported that surface modification with collagen or gelatin improved the hydrophilicity, water uptake and good mechanical strength [113]. Roskies et al. created polyetheretherketone scaffolds using Selective Laser Sintering technique with a computer-aided design program. They evaluated that PEEK scaffolds maintain the viability of adipose and bone marrow-derived MSCs and induce the osteodifferentiation of the adipose-derived MSCs [114]. Feng et al. created highly interconnected porous scaffolds with β-TCP doping of zinc oxide powder using Selective Laser Sintering technique. They studied that porous scaffold resulted in excellent mechanical and biological properties by evaluating fracture toughness, compressive strength, osteoinduction and osteoconduction [115]. Shuai et al. developed poly (vinyl alcohol)/calcium silicate composite scaffolds with interconnected porous structures and customized shapes using Selective Laser Sintering. They found excellent compressive strength, good bioactivity and cytocompatibility in these scaffolds [116]. The main advantages of Selective Laser Sintering are a wide range of materials, good mechanical strength, relatively high precision, high porosity, support materials not required, and the disadvantages of selective laser sintering are materials in powder form, difficult to remove trapped materials, expensive equipment and rough surface.
2.4. Inkjet 3D Printing
Inkjet 3D printing technique is a rapid prototyping method, which is layered manufacturing technology for creating objects described by 3D modeling software and scan data [117]. Inkjet three-dimensional printing technique is similar to Inkjet head printing. In addition, Inkjet 3D printing method has the potential to use polymeric bio-inks for various applications such as Biomedical, Tissue engineering and all other Medical fields [118-120]. Nowadays, Inkjet bioprinter is a commonly used technology, which is useful for both non-biological and biological applications. Moreover, Inkjet bioprinter is a powerful technique for depositing cells, biomaterials and has become popular in creating cell-laden constructs, which can mimic the high complexity of native tissue and organ. A typical schematic of Inkjet bioprinting is shown in Scheme 5. Many scientists were investigated using Inkjet 3D printing in tissue and organ engineering applications. Lorber et al. fabricated Retinal Ganglion Cells (RGC) and glia using piezoelectric inkjet printing. They found that the viability and survival/growth of the cells in culture were not affected by the inkjet printing process [121]. Pati et al. printed dome-shaped adipose tissue using human decellularized adipose tissue matrix bio-ink, which encapsulates human adipose tissue-derived mesenchymal stem cells from the biomimetic approach. They evaluated the efficacy of their printed tissue constructs for adipose tissue regeneration [122]. Irvine et al. created a patterned 3D structure using Inkjet bioprinter and used printable gelatin as an ideal material crosslinked with microbial transglutaminase to print cell bearing hydrogel for three-dimensional constructs. They confirmed excellent cell affinity [123]. The main advantages of Inkjet 3D printing in tissue engineering applications are cells, tissues and hydrogel that can be printed, patient-customized fabrication, incorporation of drug and biomolecules, low cost and rapid production, and the disadvantages in Inkjet printing are a limitation of size, low resolution, low mechanical properties, using limited biomaterials.
CONCLUSION AND FUTURE DIRECTION
Rapid intellectual shine in the material science field uses nanoscopic materials for various societal applications: Nano Electronics, Tissue Engineering, Artificial Intelligence, etc. through various printing methodologies. SLA can be able to produce a high resolution of excellent surface finish products in a short period of time. The capability of using multiple prints heads provides choice to select different colors and materials on making the biomedical 3D products using FDM. SLS has the functionality to make highly complex geometry with more accuracy. Usage of bio-ink through ink jet printing open doors for a biomedical researcher for producing living cell products to satisfy the need of the rare species and other living organism. Biomedical researchers keep on working in Regenerative medicine. Self-healing property, natural Self-assembly of nanotubes and synergetic effect are the key factors in the artificial bio parts manufacturing. Natural Self-assembling property of the biocompatible peptide nanotubes can be able to reform its shape if the occurred damages in the 3D printed bio Parts are under the acceptable range of damages in biostructure like bone, tissues etc. This technique can be able to reduce the number of failure in the final products and minimize the requirement of design, production and overall cost. The precise designing using the Biomedical CAD software for customized design of tissue and organs for biomedical application, which can design the required 3D model. To manufacture the designed product, a unique combination of cells with the biopolymers can act as the core material for organs and tissues. The recent development of rapid prototyping process leads the manufacturing of living tissues and organs with highly porosity architecture. To obtain a highly efficient manufacturing model, the multifunctional 3D structure is undergoing recent development by combining more than two 3D printing technology or combination of 3D printing technology with other scaffold 3D printing technology. The quality of the functioning of the scaffold is mainly due to the integrity of design, materials and the manufacturing process. Improving biomaterials quality (Polymers and Bio-ink) is the most significant goal in Additive Manufacturing, which should be biocompatible, ease of processing, good mechanical properties for cell support and secure 3D structure. 3D printing also used for drug delivery, Chemical, Biological agents and Organ on-chip devices along with tissue engineering. The scientific community is now improving the resolution, speed of prototyping and quality of printing process by compatible with cells and tissues in 3D manufacturing. Patient customized 3D bioprinting is still a challengeable process of implementing for the whole global community. Moreover, the mimicking ability of the direct organ fabrication in the medical field is the goal of tissue engineering and regenerative medicine using 3D bioprinting. Fabrication of the scaffold for the mass bone defect is the crucial process in the 3D manufacturing due to unsatisfactory bone graft substitutes. The capability of integrating various disciplines: chemistry, materials science, computer-aided design, medical imaging and biomedical world should focus on to improve the availability of various models for different patient specific applications [124-126].
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
One of the authors, M. Gundhavi Devi would like to thank Prof. Insup Noh, Convergence institute of Biomedical and Biomaterials department, SEOUL TECH, Seoul, Korea for his valuable motivation to work on 3D-bioprinting.


