All published articles of this journal are available on ScienceDirect.
Biosurfactant Production and Biodegradation of Leather Dust from Tannery
Abstract
Background:
The leather industry contributes vast amounts of pollution damaging to aquatic and terrestrial environments. Leather dust is a chromium-contaminated waste produced from the shaving and buffering processes involved in leather tanning. Microorganisms have been investigated for their usefulness in bioremediation and recycling of waste materials. Solid leather waste is the current focus of material to be remediated in this study.
Objective:
The present work focuses on the development of a process to degrade the leather dust protein with the aim of removing the chromium bound within the protein. As part of the study, detecting the presence of biosurfactant production was performed to fuel further interest in value-added by-products of the process.
Method:
Bacillus subtilis SA-6 was used to treat the leather dust over a 10 day shake flask study. Daily samples were taken and analysed for chromium content by Atomic Absorption Spectrometry. The surface tension of the shake flask cultures was also investigated to detect for any valuable by-products such as biosurfactants for future prospects of developing an economically viable process.
Results:
Chromium concentration demonstrated an exponential increase between 0-120 h in shake flask experiments. In the presence of B. subtilis SA-6 chromium concentration in cell free supernatant increased from 0.13±0.09 mg/L to 190.81±20.18 mg/L compared to when B. subtilis SA-6 was absent. Surface tension decreased during fermentation from 53.23±0.92 mN/m to 30.13±0.15 mN/m in 24 h.
Conclusion:
This study demonstrates a waste management process, which detoxifies solid tannery waste to reduce environmental pollution, whilst yielding value-added products (such as biosurfactant) to provide an economically viable bioprocess with potential for large-scale development.
1. INTRODUCTION
Leather manufacturing is a traditional industry which contributes greatly to the global economy and is one of the largest industries in the world due to the demand and versatility of leather [1]. The leather processing converts skin of dead animals such as cattle into hide through a series of chemical and biochemical treatments. In a United Nations Industry Development Organisation (UNIDO) report in 2010, the value of the leather exported from Asian countries had an estimated value of €3,710 million in 2003 [2].
Despite the great economic value and importance of the leather industry, it carries a great burden by contributing vast amounts of pollution damaging the environment. The stages of leather processing involve a wide range of chemicals which are used to convert and treat raw skin hides into commercially valuable leather. This generates enormous amounts of liquid and solid wastes. Poor management of these wastes results in severe pollution issues and reduces quality of ecological and social environments [1, 3-5].
Chromium salts are one of the chemicals utilised in the tanning process of leather to stabilise and strengthen the material [6]. Chromium is a heavy metal and although found in traces naturally in organisms, at critical levels it can cause serveral health defects affecting the central nervous system and due to its oxidising activity it is classed as a carcinogen [3]. Chromium can be found in the solid waste material produced from shaving, buffering and trimming leather [4]. Due to its toxicity, chromium is difficult to manage as waste and is becoming a high priority in the industry. The production of solid wastes contaminated with chromium is of growing concern as current methods for waste removal includes landfill and incineration, which are costly and have severe impact on the environment due to leaching of chromium ions and generation of toxic emissions including nitric oxide [1, 7, 8]. Also, the accumulation of small leather particles in the form of dust causes considerable health issues for employees and communities surrounding factories, as the inhalation of chromium-containing leather dust particles may result in cancer, respiratory diseases and central nervous system abnormalities [1, 9].
The use of biological tools is an approaching decontamination method to treat chromium contaminated wastes instead of prevalent chemical methods, which may contribute secondary pollutants. Microorganisms are being investigated for their usefulness in waste water and solid waste treatments to reduce or remove the pollutant with the potential to recycle or reuse the waste materials [10]. There have been examples where species of microorganisms have been utilised to remediate contaminated wastes of heavy metals such as, bacterial consortium treatment of contaminated soils, waste degradation by microbial attack and effluent treatment through biofilm filters [5, 10-14].
Removal of chromium bound within the collagen matrix of the leather is vital to reduce the toxicity of the material. Whilst chromium is bound within the collagen matrix, dust particles produced from shaving and buffering processes may cause occupation health risks through inhalation. Also, chromium leaching may occur through landfill waste disposal [15]. To release chromium from the matrix, the bonds between chromium and collagen need to be broken. This may be achieved through bacterial treatment for the degradation of the leather by proteases [7]. The use microorganisms in the waste management of chromium-contaminated solids could also generate a process that contributes economically valuable by-products. This includes biosurfactants produced by B. subtilis,which are being discussed in this research.
The current research investigates an environmentally friendly approach; using bacterial species to manage the solid chromium waste produced from the tanning industry. Studies into microbial degradation of leather have been carried out previously using various species of microorganisms [7, 15]. The research aims to use B. subtilis to degrade and detoxify the waste material, leather dust, a product from the buffering, shaving and trimming processes in tanning.
Removal of chromium from the collagen matrix of leather dust will reduce the toxicity of the material contributing to the elimination of occupational risk whilst providing value by-products.
2. MATERIALS AND METHODS
Leather dust (also known as chrome shavings or chrome buffering dust) was provided by a private party and donated to the project by a Teesside University Academic Supervisor. The material had been treated by a chrome tanning process and is the waste of shaving and buffering processing.
2.1. Microorganisms and Culture Conditions
B. subtilis SA-6 was cultured from existing Nutrient Agar (Oxoid, Hampshire, England) plate culture provided by the laboratory to a freshly prepared Nutrient Agar plate. Strain SA-6 was originally isolated from oil contaminated sample as a part of ongoing research programme at Teesside University and 16S rRNA identification was used to confirm Bacillus subtilis. The streak plate method was applied under aseptic conditions. Inoculated Nutrient Agar was incubated at 30°C for 24 h. The growth of B. subtilis SA-6 on Nutrient Agar was used to inoculate 150 mL Nutrient Broth (Oxoid CM001, Hampshire, England) [16, 17] in a 250 mL conical flask by swabbing a B. subtilis SA-6 colony, using a sterilised inoculum loop and mixing the loop in the fresh liquid Nutrient Broth. The inoculated liquid broth was incubated at 30°C for 24 h at 150 rpm. Optical density was recorded after 24 h at 600nm with a resulting absorbency measured at 1.36 OD 600nm. 1% inoculum was used for experimental groups using B. subtilis SA-6 for Leather Dust degradation. All media and materials were autoclaved at 121°C for 15 minutes prior to bacteria inoculation.
2.2. Detoxification of Leather Dust Assay
B. subtilis SA-6 was cultured to investigate the detoxification of chromium-containing leather dust from the tanning industry. A series of experimental cultures were designed for the assay. Cultures were grouped as follows; Nutrient Broth (NB); NB+B. subtilis (NBBS); NB+Chromium (NBCR); NB+Leather dust (NBLD); NB+B. subtilis+Chromium (NBBSCR); NB+B. subtilis+Leather dust (NBBSLD) and NB+B. subtilis+Chromium+Leather dust (NBBSCRLD). Experimental run was for 10 days at 30°C at 150 rpm and all groups were in triplicate. Daily (every 24 h) 10 mL samples were taken aseptically over the 10 day assay for analysis (samples were unable to be taken at 72 h and 96 h due to laboratory closures). Samples were centrifuged at 4000 rpm for 30 minutes at 24°C to separate biomass and liquid culture and stored at -80°C until analysis.
2.2.1. Preparation of Leather Dust
For the experimental groups containing leather dust to investigate its detoxification by B. subtilis SA-6, 1.5 g of untreated leather dust was weighed out for each culture and separately folded in foil envelopes ready for autoclaving. This accounted for 1% of total working volume [7]. Leather dust was autoclaved at 121°C for 15 minutes. The reason for sterilisation was due to results collected from trial experimental run, in which non-sterilised leather dust demonstrated microbial contamination in control groups. Also, in previous studies autoclaving leather dust has demonstrated to prepare the leather to be more readily attacked by B. subtilis strains for degradation [7]. This may be due to the thermal denaturation of the leather structure forming proteins which are degraded by B. subtilis such as gelatin [7, 15].
2.2.2. Chromium Standard
Chromium and Nutrient Broth control was prepared to show B. subtilis SA-6 growth in the presence of chromium. 0.3849 g of Chromium (III) Nitrate.9H2O salt (BDH Chemicals, Poole, England) was dissolved in 50 mL in 0.5 M Nitric acid (Fisher, Loughborough, England). This made 1000 ppm chromium solution which was further diluted to make 100 ppm. For final dilution 1 ppm of chromium solution was used in the investigation. The final concentration was prepared to ensure detection during Atomic Absorption analysis. 1.5 mL of chromium standard solution was added to liquid Nutrient Broth. The pH was adjusted to 7 using 0.5 M Sodium Hydroxide (Acros Organics, New Jersey, USA).
2.3. Testing for Indication of Biosurfactant
2.3.1. Surface Tension Measurements
Reduction in surface tension of a liquid is an indication of the presence of a surfactant and demonstrates its surface properties [18, 19]. B. subtilis is known to synthesise a class of biosurfactants called lipopeptides [20]. Therefore in the current investigation surface tension was recorded to test for any indication of biosurfactant that may be produced in the experimental groups. Cell free supernatant was tested for surface tension measurement using the Tensiometer K9 (Krüss, Hamburg, Germany) incorporating the Nouy ring method [21]. 9 mL was used for surface tension measurement and Nutrient Broth was used as a control.
2.3.2. Drop Collapse Test
The drop collapse method was used to indicate the presence of biosurfactant in the experimental cultures. The cell free supernatant was used during this investigation. 200 µL of crude oil was evenly spread over the base of a sterile petri dish. 5 µL of cell free supernatant was slowly pipetted onto the surface of the crude oil so that a droplet was formed [21]. The drop was observed for 1 minute [22]. If the drop flattened it was given a positive result and if the drop remained domed a negative result was assigned. The diameter of droplet was also recorded to determine whether the samples were larger in diameter in relation to the control of Nutrient Broth alone, as this would also be a positive result [21]. A commercial surfactant was used as a positive control to demonstrate the expected positive result for the drop collapse method. Droplet samples containing biosurfactant are predicted to collapse, whereas samples without biosurfactant will remain domed [23].
2.4. Scanning Electron Microscopy
Pellets of cell biomass from centrifugation of culture samples from the detoxification and degradation of leather dust cultures were prepared for analysis using scanning electron microscopy (SEM). The purpose of this analysis was to investigate how chromium interacts with B. subtilis SA-6. Pellets prepared were those collected from the following experimental groups; Nutrient Broth+B. subtilis (NBBS), Nutrient Broth+Chromium (NBCR), Nutrient Broth+Leather dust (NBLD) and Nutrient Broth+B. subtilis+Leather dust (NBBSLD) at final sampling time 216 h. 200 µL of molecular grade water was added to the pellets to resuspend the material. The resuspended pellet was then pipetted onto a sterile petri dish and dried at 37°C for 72 h [24]. Adhesive 15 mm diameter posts were used to print sample onto a suitable surface for SEM analysis and gain stronger signals. SEM images were taken using the S-3400 N Scanning Electron Microscope, Hitachi. Area of interest for elemental composition was analysed using INCA Point & ID software.
2.5. Quantification of Chromium Concentration in Supernatant
Atomic Absorption Spectrometry (AAS) was applied to quantify the concentration of chromium from the cell free supernatant of samples collected from detoxification and degradation of leather dust assay [17, 25, 26]. Samples were prepared as a 1 in 10 dilution in 0.2 M Nitric acid to ensure detection within the limits of the AAS equipment. If samples were still reading concentrations too high for the equipment they were further diluted. The AAS equipment was calibrated with three chromium standards with the following concentrations; 2.5 mg/L, 5.0 mg/L and 10.0 mg/L. Nutrient Broth was used as a blank. AAS model was the Thermo Scientific iCE 300, Series AA Spectrometer and computer programme for data recording was the ThermoSOLAAR software. Method set for AAS was as follows; wavelength: 357.9 nm, measurement time of 30 sec, flame height was 10.2 mm, burner of 50 mm, gas flow of 1.1 L/min.
2.6. Chromium Quantification of Leather by Acid Digest and AAS
The concentration of chromium within the leather dust prior, to treatment with B. subtilis SA-6 was established using microwave digestion to extract the chromium and then quantified using AAS. 0.5 g of autoclaved leather dust was weighed and added to a clean microwave digestion vessel, with 10 mL of 35% (8M) Nitric Acid. This was done in triplicate and a blank was prepared containing only 10 mL of 35% (8M) Nitric Acid. Samples were run on a General Method on the Microwave Digester as follows:
Stage 1 was ran for 15 min, with temperature increasing from room temperature to 200°C, power at 1500 W then held for 20 min; Stage 2 consisted of cooling time for 35-50 min to 110°C, power at 1200 W; finally at Stage 3 temperature was cooled to room temperature between 50-70 min.
Samples were then left for 20-30 min or until no gas was being produced from the sample. After acid digestion, the samples were prepared for AAS analysis for chromium quantification as previously described.
2.7. Static Incubation
To test whether chromium release into the supernatant is influenced by the shaking and aeration caused by potential mechanical degradation from the shake incubation, a static control was designed. 1.5g of autoclaved leather dust was added to either deionised water or nutrient broth with a working volume of 150 mL in 250 mL conical flask. Triplicates for both groups were performed. Cultures were placed in a static 30°C incubator. 10 mL samples were taken every 24 h with initial sample at 0 h and final sample at 216 h. Samples were centrifuged at 4000 rpm for 30 minutes to remove pellet. The supernatant of each sample was analysed for chromium concentration using AAS as previously described.
2.8. Statistical Analysis
Each experiment was set up in triplicate. Data collected was analysed and tested to calculate average of triplicate data and standard deviation of the sample represented graphically by error bars. Means for initial rate of reaction between 0-24 h were compared by performing one-way Analysis of Variance and Dunnett test at 95% confidence interval.
3. RESULTS AND DISCUSSION
3.1. Leather Dust Biodegradation
3.1.1. Quantification of Chromium in Leather Dust
Leather dust which was untreated with B. subtilis SA-6 was tested for the chromium content using microwave acid digestion, with nitric acid and quantified using atomic absorption spectrometry (AAS). After analysis of three 0.5 g samples of autoclaved leather dust, the average concentration of chromium was 170.94±15.91 mg/L with an average of 8.547 mg per 0.5 g of leather dust roughly making up 1.7% of the material composition. Afterwards, samples of leather dust underwent 10 day (216 h) fermentation with B. subtilis SA-6 to detoxify the leather of chromium. Samples were collected on a daily basis for analysis. Optical density of cultures and chromium concentration, pH and surface tension of cell free supernatants were monitored and analysed.
3.1.2. Chromium Concentration of Cell Free Supernatant
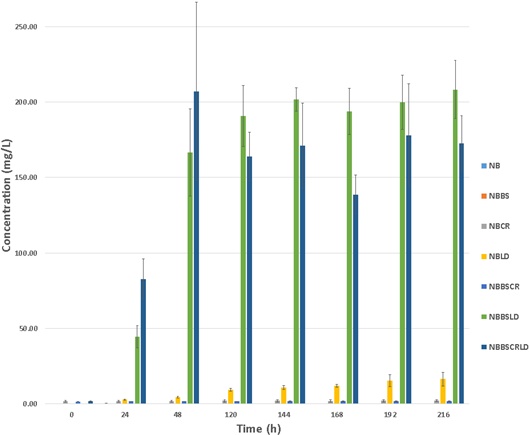
It was observed from the data collected, that chromium was released from the leather dust into the supernatant, through the action of bacterial degradation by B. subtilis SA-6. As shown in Fig. (1), with time (0-120 h), the concentration of chromium in the cell free supernatant increased when leather dust was exposed to B. subtilis SA-6. Using AAS to record chromium release, cultures NBBSLD and NBBSCRLD demonstrated the greatest increase of chromium concentration, in the cell free supernatant, between 24-48 h. NBBSLD increased from 44.4±7.32 mg/L to 166±29.01 mg/L and NBBSCRLD changed from 82.7±13.27 mg/L to 206.9±59.18 mg/L (Table 1). Post 120 h the chromium concentration in the two cultures showed no more significant changes for the remainder of the fermentation, maintaining stable concentrations. NBBSLD and NBBSCRLD fluctuated between 190.81±20.18 mg/L to 208.15±19.28 mg/L and 163.92±16.13 mg/L to 177.91±34.02 mg/L respectively. Fluctuations may be due to data collection of triplicate samples demonstrating similar trends but variation in rate of activity within cultures or in distribution of chromium in the leather dust [11].
| Time (h) | Chromium concentration (mg/L) | ||||||
|---|---|---|---|---|---|---|---|
| NB | NBBS | NBCR | NBLD | NBBSCR | NBBSLD | NBBSCRLD | |
| 0 | 0.18±0.27 | 0.03±0.03 | 1.72±0.67 | 0.11±0.03 | 1.55±0.02 | 0.13±0.09 | 1.59±0.24 |
| 24 | 0.23±0.29 | 0.03±0.03 | 1.84±0.62 | 2.74±0.33 | 1.61±0.08 | 44.41±7.32 | 82.71±13.28 |
| 48 | 0.14±0.16 | 0.04±0.02 | 1.89±0.55 | 4.44±0.52 | 1.57±0.04 | 166.61±29.01 | 206.87±59.18 |
| 120 | 0.05±0.02 | 0.04±0.03 | 1.99±0.70 | 9.34±0.94 | 1.67±0.06 | 190.81±20.18 | 163.93±16.13 |
| 144 | 0.08±0.07 | 0.03±0.03 | 1.95±0.68 | 10.80±1.19 | 1.72±0.07 | 201.79±7.81 | 171.04±28.29 |
| 168 | 0.09±0.08 | 0.03±0.03 | 1.94±0.64 | 11.97±0.96 | 1.71±0.07 | 193.78±15.44 | 138.65±12.96 |
| 192 | 0.11±0.10 | 0.04±0.03 | 2.00±0.67 | 15.26±3.97 | 1.76±0.06 | 199.89±17.92 | 177.91±34.02 |
| 216 | 0.17±0.19 | 0.03±0.04 | 2.10±0.71 | 16.42±4.52 | 1.69±0.25 | 208.15±19.28 | 172.41±18.58 |
Data collected post 120 h suggests the bioprocess of bacterial degradation of leather dust to detoxify it of chromium was completed at 120 h. The release of chromium into the supernatant is a reflection of the degradation of leather dust. Previous studies have demonstrated a similar result in the degradation of chrome tanned waste and found degradation to be complete at 120 h using Alcaligenes odorans [27]. This demonstrates B. subtilis SA-6 is as efficient as other microorganisms and can be considered a good candidate for the bioremediation of waste leather dust. The efficiency is further supported by the total chromium released from the leather dust during B. subtilis SA-6, degradation activity is found to be the same as or higher than the chromium concentration determined in leather dust through acid digestion. The average chromium concentration found in untreated autoclaved leather dust was 170.94±15.91 mg/L. From the chromium readings collected during the fermentation of leather dust and B. subtilis SA-6, there was detection of concentrations greater than 170.94±15.91 mg/L extracted from the acid digestion such as 208.15±19.28 mg/L recorded for the average chromium concentration for NBBSLD at 216 h (Fig. 1). Therefore, the aim to detoxify the leather dust of chromium was achieved in this experiment.
Control experimental groups (NB, NBBS, NBCR and NBBSCR) demonstrated negligible changes in chromium concentration over 216 h, with the highest chromium concentration detected at 48 h for NB NBBS as 0.14±0.16 mg/L and 0.04±0.02 mg/L respectively (Table 1). Differences in chromium concentration for control experimental groups containing the prepared chromium standard solution (NBCR and NBBSCR) were observed during the investigation (Supplementary document). The average chromium concentration for NBCR remained between 2.10±0.71 and 1.7±0.67 mg/L and for NBBSCR the concentration remained between, 1.76±0.06 and 1.55±0.02 mg/L. 100 ppm chromium stock solution was prepared to obtain a 1 ppm in appropriate cultures, however variation occurred and this may be accounted for by inaccuracies in pipetting instruments. Despite variation within the experimental groups, the initial concentrations for fermentation were within the chromium tolerance levels for B. subtilis [7] and were suitable for detection limits on AAS equipment.
An increase in chromium concentration was observed from control group NBLD (Fig. 1). The concentration increased from 0.109 mg/L to 16.417 mg/L between 0-216 h. This culture was not treated by the addition of B. subtilis SA-6 and the optical density shows no indication of bacterial growth by the absorption recorded (Fig. 3). The denaturing of leather dust by autoclaving may also make the leather more susceptible to disintegration whilst being shaken during incubation aiding the release of chromium. Data collected from a static incubation of deionized water and leather was compared with the data collected from a shake incubation of NBLD. It was observed that there is a greater increase of chromium into the supernatant when the culture is shaken at 150 rpm, compared to a static incubation. The final concentration of chromium for the shaken sample was 16.42±4.52 mg/L at 216 h, whereas the static sample was 1.76±0.29 mg/L suggesting shaking contributes to material break down.
3.1.3. Statistical Analysis
By studying Fig. (1), it was observed that between 0-24 h and 24-48 h there was evidence of a reaction taking place within some of the experimental groups, by the occurrence of an increase in the chromium concentration in the cell free supernatant for NBLD, NBBDLD and NBBSCRLD. Therefore, to test for any significance within the data, the rate of chromium concentration increase between 0-24 h and 24-48 h as a change in chromium concentration over time (mg/L h-1) was tested using one-way Analysis of Variance (ANOVA) at 95% confidence interval (Supplementary document).
The one-way ANOVA demonstrated a significant difference in the rate of chromium concentration increase between experimental groups, as the significance probability was <0.05 (Supplementary document). The null hypothesis of there being no significant difference in the rate of chromium release was rejected as there was no evidence to support the null as the ANOVA analysis showed significant differences between groups. This then allowed a post hoc test to be performed to distinguish which groups varied from one another. The Dunett test demonstrated a significant difference between the control group NBLD compared to NBBSLD and NBBSCRLD, with significance of probabilities <0.05 (Supplementary document).
Analysing the rate between 0-24 h and 24-48 h using the ANOVA and Dunett test statistical analysis showed a significant difference between the concentrations of chromium released over time between experimental groups. This demonstrates the addition of B. subtilis SA-6 significantly contributes the rate of chromium removal over a prolonged fermentation, to degrade and detoxify leather dust.
3.2. Growth and Activity of B. subtilis SA-6
3.2.1. Optical Density and Leather Dust Degradation
Nutrient broth was the basis of the medium for the cultures designed in this investigation for growth of B. subtilis SA-6. The optical density was measured for all samples every 24 h at 600 nm and the recorded data reflected efficient growth of B. subtilis SA-6 (Fig. 3) in this choice of media. Nutrient broth has been used as a medium choice for growth of Bacillus species, in preparation for seed culture of a continuous flow reactor bioprocess for the removal of Cr(III), using a consortium biofilm of Bacillus species [17]. The OD for NBBS cultures peaks between 24-48 h at around 30 h then begins a decline in OD readings to 120 h where the OD remains stable. The exponential phase is shown to be within the initial 24 h of incubation and the final static or death phase occurred post 48 h. Growth of B. subtilis SA-6 was only observed in repeat 3 of the NBBSCR triplicate. This resulted in OD of this experimental group to be significantly lower than other groups; with B. subtilis SA-6 present (Fig. 3). It is unclear as to why there was no bacterial growth in these cultures as B. subtilis has been observed to grow at concentrations above that used in this investigation, with growth detected at 35 ppm and 350 ppm of Cr(III) and Cr(VI) respectively [7]. Also, it can be observed from this investigation, that B. subtilis SA-6 can grow in higher chromium concentrations than the chosen standard, as it successfully degraded leather dust which was shown to have concentrations of chromium of 170.94±15.91 mg/L. Therefore, growth of bacteria was predicted for B. subtilis SA-6 in 1 ppm control solution of chromium. An unanticipated increase in average OD600 was seen in NB and NBCR groups which were affected by the variation of the triplicate shake flasks caused by contamination.
The optical density of cultures involving the degradation of leather dust by B. subtilis SA-6 (NBBSLD and NBBSCRLD), demonstrated the highest readings for all experimental groups due to the combination of the degradation of leather dust and bacterial growth with the highest recorded average OD at 192 h of 3.90±0.12 600nm for NBBSLD. Also as the leather is degraded it is dispersed within the liquid culture, affecting turbidity and increasing the absorbency of the liquid. The chromium present in NBBSCRLD did not affect bacterial growth. Cultures with leather dust and B. subtilis SA-6 present changed from the yellow transparent Nutrient broth colour to a blue/brown cloudy liquid. The blue colour is an indication of chromium released from the leather and the leather has been degraded. After centrifugation the blue colouring remained to be present in the cell free supernatant as further indication of chromium present in the sample. Similar observations have been recorded in previous studies such as Shanthi et al. (2013) where this event was seen at 48 h after incubation of leather with Alcaligenes faecalis. No settling of leather dust particles was observed after 48 h in bacterial and leather dust cultures suggesting complete liquefaction of the leather dust waste. At this stage it can be suggested up to 90% of the leather dust has been degraded as in Pillai and Archana (2012) study, where 90% of chrome shavings were degraded after 48 h of fermentation.
Bacterial growth may have been promoted by additional nutrition provided by the leather dust which would contribute to the higher OD readings recorded for the cultures with leather dust and B. subtilis SA-6. Gelatin has been recovered from animal skins which have undergone liming or tanning as a valuable product [15, 28]. The presence of gelatin in the tanned material may have acted as a substrate the bacteria could utilise for efficient growth. This is supported by studies adding gelatin to the media for growth of B. subtilis [29].
Gelatin was used as a component of the culture nutrients for the production of protease enzymes by B. subtilis in a study conducted by Pant et al. (2015). Bacillus species have demonstrated protease enzyme synthesis, which are successful at degrading fibrous proteins including collagen and keratin [30, 31]. Autoclaving the leather dust prior to treatment with B. subtilis, denatured bonds within the tightly woven structure of the triple helix of collagen, and enabled efficient attack of the leather dust by the protease enzymes produced by the bacteria [15]. The presence of gelatin in the leather may have enhanced bacterial growth thus production of proteases by B. subtilis SA-6 and the degradation of leather dust represented by the high OD readings shown in Fig. (3) [29]. Protease enzymes are also an emerging mechanism for the removal of hair on the pre-treated animal hide [7, 30, 31]. In this investigation B. subtilis SA-6 demonstrated to degrade the leather dust reflected by the release of chromium. The protein degradation of leather dust was induced by the protease activity of B. subtilis SA-6. The protease production during the fermentation process adds economic value to the management of chromium-contaminated solid waste, as the bioprocess holds the potential to produce protease enzymes which can be utilised further upstream of the leather production industry. Tanneries could produce their own in-house enzymes reducing cost of buying external protease supplies [7].
B. subtilis tolerance and/or resistance to chromium has been observed in a study conducted by Pillai and Archana (2012), in which the strain B. subtilis P13 demonstrated to be tolerant/resistant to 35 ppm of Cr(VI) and 350 ppm Cr(III) with growth detected at these chromium levels. In the current investigation growth of B. subtilis SA-6 was detected during the initial stage of degradation with OD readings and levels of chromium in the supernatant, both demonstrating a positive linear trend between 0-48 h (Figs. 1 and 3). This suggests the growth of B. subtilis SA-6 is not affected by the release of chromium. The bacterial stationary phase occurs when the degradation of leather dust is complete and there is little change in the chromium concentration post 120 h. At this stage the bacteria may have exhausted the nutrients in the broth but the process was completed as the chromium concentration in the supernatant, reflects similar concentrations found in the leather dust detected during the acid digestion and is not a reflection of intolerance to chromium.
To develop the investigation further and for potential use as a large scale bioprocess for the waste management of solid chromium-contaminated leather dust, the rate of degradation requires further investigating. The degradation of the leather dust and the release of chromium is a reflection of a kinetic reaction between the degrading protease enzyme produce by B. subtilis SA-6 and the substrate, which in this case was the leather dust. To optimise the process of degradation variations in the concentration of enzyme present and the mass of substrate should be investigated by changing the volume of seed culture or weight of leather dust. The 1% inoculum of B. subtilis SA-6 demonstrated to be successful in the degradation of 1.5 g (1% w/v) leather dust in 120 h in the current investigation, which is also presented in other research. 1% of chrome shavings have been demonstrated to be reduced by 90% over 24 h by B. subtilis P13 [7]. For a large scale process, larger volumes and high rates of reaction are necessary for the design of a sustainable and economical system. Efficiency of the degradation of leather dust by B. subtilis SA-6 should be tested by increasing the weight of leather dust. In an investigation by Pillai and Archana (2012) 5% weight of chrome shavings was suggested as the substrate concentration for degradation by B. subtilis P13 (0.6 OD 600nm), with 90% of the chrome shavings being degraded in 48 h and showed that at higher substrate percentage (10%), the efficiency of degradation was significantly reduced with less than 40% of chrome shavings degraded in 48 h [7].
3.2.2. Bacterial Interaction with Chromium
B. subtilis is a microorganism which has demonstrated biosorption of heavy metals including chromium and has been used to remove chromium as a pollutant [32]. It has proven to grow successfully in a range of chromium concentrations with ranges between 5-50 ppm [32] and was able to tolerate the chromium exposure from the leather dust in this investigation, supported by the change in optical density over time representing bacterial growth, the successful degradation of leather dust and other metabolic activity including biosurfactant production (Fig. 2). However, in this investigation there was no evidence collected to determine whether chromium was interacting with B. subtilis SA-6, through biosorption or any other mechanism of removal such as bioaccumulation. The level of chromium within the cell free supernatant post 120 h showed no further increase, or significant decrease which suggests there was no removal of chromium from the supernatant via biosorption. Results from the SEM elemental composition showed no evidence or chromium detection on the bacterial cell surface. The analysis produced images of individual B. subtilis SA-6 cells and of the leather dust and the surface of the samples were tested for their elemental composition. From the analysis of the cell free supernatant, chromium was observed to be released into the liquid over time (Fig. 1) but it was also of interest to observe if there were any direct bacterial interactions with the chromium. When samples from this investigation were tested for their chromium composition on the cell surface; either traces of chromium were detected or none at all. Traces were detected in NBLD and NBBSCR (1.42% and 0.98% respectively). No detection of chromium was recorded on the surface of B. subtilis SA-6 cells. A study carried out by Mangaiyarkarasi et al. (2011) to investigate the reduction of Cr(VI) by B. subtilis showed evidence of chromium reduction on the cell surface, demonstrating the role of membrane bound enzymes and identified the chromate reductase enzyme is produced by B. subtilis [24]. Over time the concentration of Cr(VI) decreased in the culture in their study. This was not evident in the current study.

The speciation of chromium was not determined during this study; however, due to the chromium concentration remaining relatively stable during 120-216 h it may suggest that the chromium present in the culture supernatant was Cr(III), which is commonly used in the tanning process [17]. Cr(III) is not as mobile or as soluble as Cr(VI) across cellular membranes therefore, it is less likely to be passively taken by B. subtilis by biosorption; thus, unlikely to occur in this investigation [9, 33, 34]. This does not conclude Cr(III) cannot be removed by bacterial interaction, as it has been recorded that B. subtilis in consortium with Bacillus cereus, was able to remove up to 98% of Cr(III) present in a tannery effluent by adsorption onto the biofilm in a continuous flow reactor [17]. Uptake of chromium species is dependent on the pH of the conditions in which the bioprocess is occurring. In this study the pH in which both B. subtilis SA-6 and leather dust were present, ranged between pH 7.25±0.02 before treatment and pH 8.87±0.03 after treatment (Table 2). Studies in which uptake of chromium by biosorption or bioaccumulation has occurred it has been under lower pH conditions such as the uptake Cr(III) at pH 2.5 in the study carried out by Sundar et al. (2011). The bioprocess in the shake flask cultures of the current investigation were not buffered; therefore, the metabolism of living cells and the redox reactions between the liquid culture and cells will affect the final pH of the culture conditions [11]. This was observed by an increase in pH in B. subtilis SA-6 cultures from pH 7.25±0.02 to pH 8.87±0.03 between 0-216 h (Table 2). In previous studies the rate of chromium uptake by living cells has slowed due to an increase in pH from pH 2.5 to final pH 3.5 [11]. This would suggest the pH condition in the current investigation, would create an unsuitable environment for the biosorption or bioaccumulation of chromium to remove the heavy metal from the liquid culture after the release of chromium by the degradation of leather dust. This is supported by the levelling of chromium concentration post 120 h for cultures with both leather dust and B. subtilis SA-6 (Fig. 1). At higher pH there is likely to be less protons available to assist the uptake of chromium through interactions with carboxyl groups in the peptidoglycan cell wall [11, 35].
| Sample | NB | NBBS | NBCR | NBLD | NBBSCR | NBBSLD | NBBSCRLD |
|---|---|---|---|---|---|---|---|
| Time(h) | pH Average ± Standard Deviation | ||||||
| 0 | 7.25±0.01 | 7.25±0.02 | 6.96±0.03 | 7.12±0.02 | 7.05±0.02 | 7.09±0.02 | 7.02±0.03 |
| 24 | 7.45±0.07 | 8.29±0.04 | 7.18±0.01 | 6.48±0.13 | 7.64±0.71 | 7.97±0.02 | 8.31±0.03 |
| 48 | 7.30±0.15 | 8.68±0.04 | 7.16±0.01 | 6.40±0.13 | 7.69±0.90 | 8.40±0.02 | 8.61±0.04 |
| 120 | 7.39±0.03 | 9.00±0.08 | 7.15±0.03 | 6.14±0.15 | 7.73±1.11 | 8.94±0.03 | 9.02±0.01 |
| 144 | 7.23±0.01 | 8.88±0.07 | 7.07±0.01 | 6.09±0.13 | 7.70±1.09 | 8.91±0.04 | 9.02±0.03 |
| 168 | 7.60±0.54 | 8.95±0.06 | 7.13±0.02 | 6.11±0.23 | 7.69±1.06 | 8.91±0.06 | 8.97±0.03 |
| 192 | 7.71±0.70 | 8.90±0.06 | 7.14±0.01 | 6.06±0.17 | 7.72±1.09 | 8.82±0.03 | 8.94±0.03 |
| 216 | 8.07±0.65 | 8.79±0.06 | 7.67±0.91 | 6.14±0.21 | 7.69±1.01 | 8.75±0.03 | 8.87±0.03 |
Adjusting the pH post 120 h, which is the time in which degradation is considered to be complete, may enable the removal of chromium by the B. subtilis SA-6 cells. The optical density of B. subtilis SA-6 growth curve (Fig. 3) shows the bacterium reaches a death or stationary phase between 24-48 h when only present in Nutrient broth. Living cells will eventually exhaust the nutrients within the medium, but this does not reduce their potential to biosorb chromium. Both living and dead cells can be utilized for the biosorption of chromium [11]. Therefore, this experimental set-up may be designed to alter the pH post completion of leather dust degradation to then focus on biosorption of chromium, by adjusting the pH to make the conditions suitable for removal of chromium.
3.3. Exploitation of Value-added Products from Detoxification Process
3.3.1. Production of Biosurfactants by B. subtilis SA-6
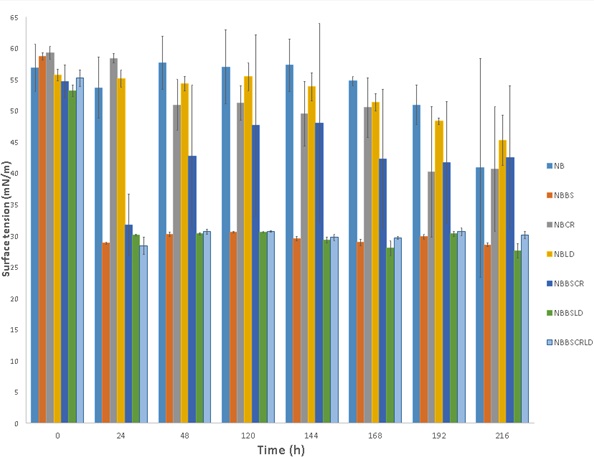
Bacillus species have been identified as producers of a large class of biosurfactants called lipopeptides, which consist of a cyclic polypeptide linked to a long fatty acid chain [20]. They can be divided into several groups which include surfactins, fengycins and iturins [20]. Following centrifugation of samples collected from the leather dust detoxification fermentation, the cell free supernatant was used to test the surface tension of the liquid for any indication of biosurfactant production. Demonstrated in Fig. (2) is the reduction in surface tension of liquid Nutrient broth when B. subtilis SA-6 is present. For culture NBBS, after 24 h the surface tension reduced from 59.3±0.6 mN/m at time 0 h to 28.8±0.2 mN/m. A reduction in surface tension is an indication of the production of biosurfactants and B. subtilis synthesises the class of biosurfactants, lipopeptides [20]. Reduction in surface tension is also observed in other cultures with B. subtilis present and includes cultures with leather dust present as well. NBBSLD and NBBSCRLD both demonstrated a reduction in surface tension by 23.1 mN/m and 26.8 mN/m respectively. This is a positive result in which biosurfactant production is occurring and the synthesis is not affected by the presence of toxic chromium from the leather dust. NBBSCR was another culture with B. subtilis SA-6 present; however, the extent of surface tension reduction was not as great as other B. subtilis SA-6 cultures. This may be in response to the lack of bacterial growth observed in these cultures.
The presence of biosurfactant was further supported by the drop collapse analysis of the cell free supernatants; Table 3 represents the data recorded for samples collected between 0-24 h containing combinations of bacteria or leather dust. Samples with B. subtilis SA-6 present in the culture showed evidence of drops collapsing on crude oil which was supported by an increase in droplet diameter. It can be observed that the diameter of the collapsed drop in NBBS and NBBSLD increased from 2.8±0.3 to 5.3±0.3 mm between 0-24 h (Table 3) whereas, the NB control remained a stable drop on the crude oil surface. This is a qualitative indication of biosurfactant production and supports the trends of surface tension represented in Fig. (2).
| Sample | Diameter of drop (mm) 0 h | Diameter of drop (mm) 24 h | ||
|---|---|---|---|---|
| Average | Standard deviation | Average | Standard deviation | |
| NB | 3.2 (-) | 0.3 | 3.1 (-) | 0.4 |
| NBBS | 2.8 (-) | 0.3 | 5.3 (+) | 0.3 |
| NBBSLD | 2.8 (-) | 0.3 | 5.5 (+) | 0 |
| Positive control | 12.5 | 19.5 | ||
Due to their amphiphilic properties, lipopeptides synthesised by B. subtilis have the potential to be used in a variety of applications which include medicinal uses, environmental applications and cosmetics [36]. Biosurfactants are of great interest due their advantageous qualities including their biodegradability being naturally derived products, unlike their chemically synthesised alternatives [37]. The lipopeptides produced by Bacillus species are considered to be powerful biosurfactants with surfactin demonstrating to reduce water surface tension by 45 mN/m [38].
The production of a biosurfactant creates additional value to the waste management process if it can be purified from the bioprocess. The surrounding environment of an industrial setting is at risk to contamination from hydrocarbons and heavy metals, with current technologies it has been difficult to develop a process which treats both simultaneously [39]. The crude biosurfactant produced by B. subtilis has been shown to remove high percentages of crude oil and motor oil from contaminated sand, with up to 88% removal [39]. It was also observed that heavy metals are removed up to 100% using the crude biosurfactant of B. subtilis which included chromium, copper and zinc [39]. Therefore, the biosurfactant produced during the leather dust and B. subtilis SA-6 fermentation may hold promise for applications for bioremediation. However, the production of lipopeptides from B. subtilis SA-6 needs to be optimised to make exploitation of the biosurfactant viable. This may be achieved by the correct medium composition for optimal growth conditions, for example, glycerol in medium has shown to give high yield of lipopeptides and is an inexpensive substrate [39].
3.3.2. Further Developments and Economic Value of Bioprocess
The process of detoxifying leather dust protein by the degradation process of B. subtilis SA-6, has been observed to remove chromium from the fibrous matrix of the leather dust from this investigation. There is also evidence of value-added products which can be exploited to make this a viable process, such as biosurfactants.
There are also other added-value products which have the potential to be exploited from the degradation and detoxification of leather dust. This includes the protease enzymes produced during the degradation process, which are also being utilised further upstream in the leather processing industry to remove hair on untreated animal hide. Also, with the removal of chromium from the collagen protein which is the main component of the leather dust the collagen is no longer toxic and may be utilised for the medical uses in bone graft engineering [27]. Finally, chromium itself can be recovered for nano-particle production and be reused in the chemical industries [17]. This investigation shows promise for a bioprocess that may have high economical value resulting in a viable waste management process.
CONCLUSION
In conclusion, B. subtilis SA-6 successfully degraded leather dust to detoxify the material of chromium, one of the top pollutants of main environmental and health concern. By utilising a biological method, less energy is required, there are fewer risks of secondary pollutants, than when using chemical methods [14]. Whilst performing this process there was evidence of biosurfactant production with detection of reduced surface tension of the liquid culture. This shows promise for the waste management process to be developed in the future into a large scale bioprocess, which tackles the waste pollutant and produces potential added-value by-products for an economically viable process.
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publishers Website along with the published article.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
The authors would like to thank Teesside University for the access and use of the laboratory facilities and the help and support given by technicians. Also, thanks is given to Shirley O’Hare for discussion and advice during the project and Chibuzo Uzoigwe for guidance on biosurfactant testing techniques. Finally, support was given by the Postgraduate Experience Programme scholarship.


