All published articles of this journal are available on ScienceDirect.
Impact of Imidacloprid Seed Dressing Treatment on Soil Microorganisms and Enzyme Activities in the Maize Rhizosphere
Abstract
Under the field conditions, effects of imidacloprid seed dressing treatment on soil culturable microorganisms and enzyme activities in maize rhizosphere were studied. The results showed that the microbial populations for bacteria, actinomycetes and fungi in maize rhizosphere after imidacloprid treatments were lower than control. The bacteria and actinomycetes populations showed a trend of decreasing after increasing with the maize growth from the seedling stage to the maturity stage, and the fungi populations decreased with the maize growth. The urease activities of maize rhizosphere soil from different treatments showed a trend of initially increasing after decreasing, then decreasing. The invertase activities of maize rhizosphere soil from different treatments showed a trend of decreasing after increasing, and the peak value occurred at flowering stage. With the imidacloprid application, the invertase activities had been stimulated. The results may provide theoretical basis for rational seed dressing treatment.
1. INTRODUCTION
The imidacloprid, 1-[(6-chloro-3-pyridinyl)methyl]-4,5-dihydro-N-nitro-1H-imid azol-2-amine, is a nitromethylene derivative synthesized in February 1985 by Nihon Bayer Agrochem K.K [1]. Having both contact and systemic properties, it can provide protection from the time of crop seedling emergence well into the growing season [2]. The product Gaucho® (active ingredient imidacloprid)is used for seed dressing due to its prevention properties against soil and sucking insects [3]. The seed treatment formulation minimizes the amount of chemical used in the crop growth period, but it may sometimes be unnecessary as it is applied before pest attacks [4]. It has been reported that only 0.1% of the applied pesticides reached the target pests and remaining 99.9% find their way to different components of the environment [5]. In recent years, soil pollution caused by human-made chemicals is becoming more and more serious [6]. Thus it is necessary to assess pesticides’ impact on soil biochemical properties to avoid the pollution brought about by them [7]. Plant rhizosphere is micro domains in contact with the soil and it is the main area for absorption of plant nutrients [8]. Generally, soil microbes are the basic soil components and soil enzymes contribute to the total soil biological activities because they are intimately involved in organic matter decomposition, nutrient cycling, energy transfer, and environmental quality [9-11]. Moreover, soil microbes represent the unseen majority in soil and comprise a large portion of the genetic diversity on Earth [12].The soil enzyme activity and soil microorganisms are also the important indexes of soil fertility. Improving the soil enzyme activity, can promote plant growth, control and reduce the harm of diseases and pests, and ultimately increase crop yield [7, 11]. However, to the authors’ best of knowledge, under field conditions, effects of imidacloprid seed dressing treatment on soil microorganisms and enzyme activities in maize rhizosphere have not been studied. The aims of this work were to examine the impacts of imidacloprid on soil microbe population or abundance and enzyme activities. The findings are useful to understand the interactions between pesticides seed treatments and microbes in agricultural soils, and they can provide theoretical basis for rational utilization of seeds treatment pesticides.
2. EXPERIMENTAL DESIGN AND METHODS
2.1. Experimental Design
Gaucho®, 600g/L imidacloprid flowable concentrate for seed coating,purchased from Bayer crop science (China) Co., LTD. Maize hybridized combination ‘1/15’, provided by Corn Research Center of Henan Institute of Science and Technology, was used as plant material in this study. The experiment was conducted from June 7 to Ocotober 10 of 2014 in Hongmen Town of Xinxiang City, Henan Province. A field (sandy loam soil, pH 7.3) previously cultivated with wheat was used for this experiment. The soil chemical properties before the experiment were as follows: soil organic matter 9.47 g kg-1, total nitrogen 0.38 g kg-1, total phosphorus1.17 g kg-1, total potassium 6.16 g kg-1, available nitrogen 20.18 mg kg-1, available phosphorus 38.24 mg kg-1, available potassium 219.07 mg kg-1. Two different treatments were included: 1) control plots (without imidacloprid treatment, CK); 2)treated plots (seed dressing treatment at 5ml of imidacloprid per 1kg corn seed, homogeneously mix and make them dry, then sown at same space, the line space is 60 cm, the density is 90000 plants/hm2). Each treatment has three replicates (30 m2) and the study plots were completely randomized. All plots were subjected to the same fertilization and field management.
2.2. Method of Sampling
At the seedling stage, jointing stage, trumpet stage, flowering stage, filling stage and maturity stage during the maize whole growing period, the plants were chosen according to three point sampling method (10 plants were collected at one point), the rhizospheric soils tightly attached to roots were collected and then sieved through 2 mm mesh to remove plant roots, leaf remains and insects for further analyses.
2.3. Determination Method and Data Analysis
The number of was determined with serial dilution and a pour plate method [13-15]. Ten grams of soil from each sample was suspended in 100 ml of sterile water and 10-fold serially diluted. Nutrient agar medium for bacteria was beef extract-peptone medium and for actinomycetes was Gause-1 medium and for fungi was Martin’s medium containing rose bengal 33 μg/ml and streptomycin 30 μg/ml. Serial soil dilutions were prepared according to the desired selective microorganisms. The inoculated agar plates were incubated at 28±2°C for 3 days for bacteria and fungi and 7 days for actinomycetes before the colonies were counted. Results were calculated as CFU per g dry soil. The percentages of growth inhibition were calculated by the formula which was as follows:
Growth inhibition(%)=(1-T/C)×100
where C is the number of soil microorganisms in the control and T is the number of soil microorganisms in the treatments.
Urease activity was determined using urea as substrate and the soil mixture was incubated at 37°C for 5 h. The residual urea was determined by a colorimetric method. The results were presented as mg urea-N kg-1soil 5h-1 [16]. Invertase activity was determined as the method based on incubation of soil samples using a sucrose solution as substrate at 37°C for 24 h. Blanks were incubated without substrate or soils. The result was shown as mg GE (glucose equivalents) g-1soil 24 h-1 [17].
The results obtained were analyzed using Statistical Product and Service Solutions (SPSS). All reported results were means of three replications and the data were compared by Duncan’ s new multiple range test at the 5% level. Differences between values at P < 0.05 were considered statistically significant.
3. RESULTS AND ANALYSIS
3.1. Effects of Imidacloprid on Maize Rhizosphere Soil Mirobial Culturable Populations
The results showed that microbial populations for bacteria, actinomycetes and fungi in maize seed coated with imidacloprid were lower than control soil (Table 1). At the seedling stage, the bacteria populations and actinomycetes populations were inhibited by 13.54% and 6.83%, respectively. As concerned as bacteria and actinomycetes populations, there are statistically significant difference between the flowering stage and other growth stages in the control and treated samles. The number of the microbial populations showed a trend of decreasing after increasing with the maize growth from the seedling stage to the maturity stage, and their peak value occurred at the flowering stage. The peak value for bacteria and actinomycetes populations in the imidacloprid treated samples were 100.6×107CFUg-1 soil and 91.3×105CFUg-1 soil, respectively. However, the fungi populations decreased with the maize growth, and there are statistically significant difference between the seedling stage and other growth stages in the control and treated samles. and the fungi populations present an opposite trend compared with the bacteria and actinomycetes, the peak value occurred at seedling period. And in the seedling stage, the populations for fungi in the control and treated samples were 90.7×104CFUg-1 soil and 88.2×104CFUg-1 soil, respectively.
|
Microorganism (Culturable population) |
Imidacloprid | Growth stages | |||||
|---|---|---|---|---|---|---|---|
| Seedling stage | Jointing stage | Trumpet stage | Flowering stage |
Filling stage |
Maturity stage | ||
| Bacteria (107CFUg-1 soil) |
CK | 80.5±7.4f | 91.4±8.5bcd | 97.6±10.3bc | 114.7±12.6a | 100.3±11.5b | 82.6±6.9ef |
| Imidacloprid | 69.6±5.3g | 79.8±10.2f | 88.3±7.6cde | 100.6±9.3b | 95.7±6.9bc | 83.4±7.1ef | |
| Actinomycetes (105CFUg-1 soil) |
CK | 67.3±6.5f | 76.8±5.4e | 87.2±9.1bcd | 99.7±10.2a | 93.3±9.6ab | 90.3±7.8bc |
| Imidacloprid | 62.7±5.9f | 69.6±8.1f | 80.7±9.2de | 91.3±11.6bc | 89.6±7.3bc | 84.9±7.4cd | |
| Fungi (104CFUg-1 soil) |
CK | 90.7±10.2a | 86.2±6.3ab | 81.4±7.9abcd | 75.5±4.8cd | 77.9±9.2bcd | 74.8±8.6d |
| Imidacloprid | 88.2±8.5a | 84.3±7.1abc | 76.7±7.7cd | 72.3±6.9d | 73.4±5.4d | 72.9±5.3d | |
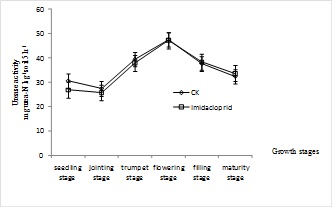
3.2. Effects of Imidacloprid on Maize Rhizosphere Soil Enzyme Activities
With the maize growth, the urease activities of maize rhizosphere soil from different treatments showed a trend of initially increasing after decreasing, then decreasing, (Fig 1). And the peak value occurred at flowering stage, the value increased by 55.74% and 76.12% in control and imidacloprid treated soils, respectively. Compared with control rhizosphere soil, urease activities in imidacloprid treated soils were inhibited from seedling stage to flowering stage, and the value decreased by 0.63%-12.13%. The invertase activities of maize rhizosphere soil from different treatments showed a trend of decreasing after increasing, and the peak value occurred at flowering stage, (Fig. 2). With the imidacloprid application, the invertase activities had been stimulated. From seedling stage to flowering stage, the value increased by 8.69%, 3.34%, 4.97% and 3.91%, respectively.
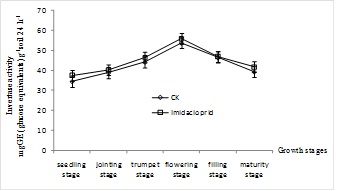
DISCUSSION
Soil enzyme activity is a form of soil biological activity, and it is also the important indicator of soil fertility levels. It can reflect the soil nutrient level in particular the transformation ability of N and P, but soil enzyme activity is also affected by soil nutrient status and soil texture [7, 11]. Many research results showed that the pesticide had dual effects on the growth of crops, both promoting effect and inhibitory effect [18, 19]. This test is to determine the key soil enzymes which affect crop growth by imidacloprid seed treatment under the same conditions. Urease is involved in the hydrolysis of urea-type substrates and its hydrolyzate urea is an important N fertilizer. Invertase involved in the carbon-cycle and it was critical to release low molecular weight sugars that were important for microorganisms. The present study implied that soil enzymes exhibited different behavior when exposed to imidacloprid.The literature shows that various studies have investigated the effects of pesticides on the soil environment. For example, in imidacloprid seed treated groundnut (Arachis hypogaea L.) field, both dehydrogenase and phosphomonoesterase activities were increased between 15 and 60 days after sowing [20]. Wang et al. used soil enzymatic activities, dehydrogenase, phosphomonoesterase, arginine deaminase, and urease to evaluate whole metabolic activity changes and acute toxicity following imidacloprid treatment under greenhouse conditions [21]. The rhizosphere soil enzyme urease, alkaline phosphatase, protease, invertase and catalase activity showed a trend of decreasing after increasing under different carbofuran imidacloprid thiram complex concentration treatments [8]. The results of this paper showed that urease activities were inhibited by imidacloprid compared with the control, and which is the same as the past work. It may be that rhizosphere microenvironment is in adversity conditions and its normal physiology is inhibited, moreover enzyme activity decreased. The results also showed that invertase activities had been stimulated by imidacloprid compared with the control. This may be due to the action between seed treatment pesticides and soil environment. Soil enzyme activity is not simply activated by pesticides, also may be affected by maize growth and root secretion, and promote the growth of microorganisms, thereby increase the soil enzyme activity.
The rhizosphere is the interface between plant roots and soil where interactions among a myriad of microorganisms affect biogeochemical cycling, plant growth [22]. Some of the microbes that inhabit this area are bacteria that are able to colonize very efficiently the roots or the rhizosphere soil of crop plants. They fulfill important functions for plant growth and health by various manners [23]. Soil microbes are the basic components of the soil ecosystem and are vital for the degradation of pollutants in soils. And soil microbes are also often sensitive to environment change and stressors. Our results also showed that microbial populations for bacteria, actinomycetes and fungi in maize seed coated with imidacloprid soil were inhibited, but with the maize growth, these microorganisms also increased. This implied that, on one hand, imidacloprid (or its metabolites) might be toxic to some microorganism species, on the other hand, microorganisms might be associated with the maize roots secrets.
The soil microbial community plays an essential role in the interactions between pesticides and microorganisms [24]. In our future study, new analytical technologies, such as phospholipid fatty acid (PLFA) analysis, polymerase chain reaction-denaturing gradient gel electrophoresis (PCR-DGGE), and physiological profiles in microplates, have greatly helped the study of the soil microbial community. Moreover, the action mechanism of imidacloprid on soil enzyme activities also should be further studied.
CONCLUSION
In this paper, impacts of imidacloprid seed dressing treatment on soil culturable microorganisms and enzyme activities in maize rhizosphere were studied under the field conditions. The bacteria and actinomycetes populations showed a trend of decreasing after increasing with the maize growth from the seedling stage to the maturity stage, and their peak value occurred at the flowering stage. The peak value for bacteria and actinomycetes populations in the imidacloprid treated samples were 100.6×107CFUg-1 soil and 91.3×105CFUg-1 soil, respectively. And the fungi populations present an opposite trend compared with the bacteria and actinomycetes, the peak value occurred at seedling period. And in the seedling stage, the populations for fungi in the control and treated samples were 90.7×104CFUg-1 soil and 88.2×104CFUg-1 soil, respectively. The urease activities of maize rhizosphere soil from different treatments showed a trend of initially increasing after decreasing, then decreasing. Compared with control rhizosphere soil, urease activities in imidacloprid treated soils were inhibited from seedling stage to flowering stage, and the value decreased by 0.63%-12.13%. The invertase activities of maize rhizosphere soil from different treatments showed a trend of decreasing after increasing, and the peak value occurred at flowering stage. From seedling stage to flowering stage, the value increased by 8.69%, 3.34%, 4.97% and 3.91%, respectively. The findings can provide theoretical basis for rational utilization of imidacloprid seed dressing treatment.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
This work was supported by National Natural Science Foundation of China (31201528) and Aid Project for the Leading Young Teachers in Henan Provincial Institutions of Higher Education of China (2013GGJS-135).


