All published articles of this journal are available on ScienceDirect.
Micropropagation of Gapi Banana (Musa sp.) using Aloe vera Gel Juice and Vigna radiata Sprout Extract as Natural Plant Growth Regulators Sources
Abstract
Introduction
The most essential factor in the micropropagation of Gapi banana plants is the induction of initial growth. The initial growth induction of cultures usually uses the growth regulators cytokinin and auxin or a combination of both, and can also utilize natural materials, such as Aloe vera and Vigna radiata, as a potential alternative to synthetic PGRs to induce shoots/callus/somatic embryos. This study aims to evaluate the effects of Aloe vera gel juice and Vigna radiata sprout extract, compared to the control media (MS-0), the BAP, and the combination of BAP with 2,4-D in Murashige and Skoog (MS) culture media in the micropropagation of Gapi banana.
Methods
The study was conducted as a laboratory experiment using Murashige and Skoog (MS) medium supplemented with Aloe vera gel juice (MS-LB) 10, 15, 20, and 25% (v/v), Vigna radiata sprout extract (MS-ET) 10, 15, 20, and 25% (v/v), and compared with control media MS-0, MS-BAP 1 mg L-1, and a combination of MS-BAP 1 mg L-1 with 2,4-D (0.1, 0.3, and 0.5 mg L-1). Data were analyzed using the Chi-square (χ2) test to determine differences among treatments.
Results
The results were that the explants could grow to form the best shoots on 20% MS-LB medium and 10% MS-ET in the second month, and 1 mg L-1 MS-BAP medium formed several shoots in the third month. Adding a combination of 1 mg L-1 BAP + 2,4-D of 0.1 and 0.3 mg L-1 in MS media produced a compact callus. High concentrations of MS-LB medium (25%), 15 and 25% MS-ET media, and a combination of 1 mg L-1 MS-BAP + 0.5 mg L-1 2,4-D medium did not support explant growth because the inoculant remained blackened and did not develop.
Discussion
The findings of this study suggest that Aloe vera gel juice and Vigna radiata sprout extract can be considered as promising natural alternatives to synthetic plant growth regulators in banana micropropagation. This approach highlights the potential of eco-friendly and cost-effective culture media supplements to support sustainable plant tissue culture practices.
Conclusion
MS media added Aloe vera gel juice (MS-LB 20%, v/v) and Vigna radiata sprout extract (MS-ET 10%, v/v) were most effective in inducing shoot growth in the second month after inoculation, faster than 1 mg L-1 BAP treatment medium, which succeeded in forming shoots in the third month. The combination of BAP 1 mg L-1 + 2,4-D 0.1 and 0.3 mg L-1 created a compact callus from the first month. On the other hand, high concentrations of 25% (v/v) MS-LB, 15 and 25% (v/v) MS-ET medium, and the combination medium of 1 mg L-1 BAP with 0.5 mg L-1 2,4-D did not support explant growth because the explant remained blackened and did not develop.
1. INTRODUCTION
Banana is one of the most important horticultural commodities with numerous benefits. Among the various types of bananas, one notable variety is 'Gapi Merah’. This banana variety was officially described in 2022 with the decree number SK.451/Kpts/PV.240/0/XII/2022, in which the author was one of the researchers involved. Banana fruit can be consumed either raw or processed. The peel and stem of the banana plant can be extracted and used as an antifungal agent for oral pathogens [1]. Banana fruit peel contains phenolic antioxidants [2], organic acids, various types of enzymes, and can be used as organic fertilizer, an energy source [3], and an adsorbent. The banana flower can be used as a vegetable, while banana leaves serve as food wrappers or ingredients for various purposes.
In general, banana propagation is traditionally done through suckers. A single banana rhizome that has been harvested can produce 2 - 4 new banana suckers, which are ready to be transplanted within 8-9 months. However, this method provides only a limited number of banana seedlings within that period, while the demand for commercial planting has grown. Therefore, an alternative method is needed to mass-propagate banana plants using non-conventional breeding techniques, specifically through plant micropropagation (in vitro culture) [4]. Banana micropropagation is pivotal for producing disease-free, high-yielding, and uniform banana plants, supporting sustainable production systems. According to FAO (2023), global banana improvement programs highlight tissue culture and mutation breeding as essential tools for varietal development, genetic improvement, and disease management. Recent progress in banana tissue culture highlights the integration of biotechnological approaches to overcome contamination and browning issues, improving large-scale propagation efficiency.
Plant micropropagation is an in vitro plant multiplication technique conducted on a laboratory scale. This technique is carried out in transparent, heat-resistant glass containers filled with a culture medium containing various nutrients/minerals and other essential components, all under aseptic/sterile conditions. Generally, micropropagation is performed to produce many plants in a relatively short period while maintaining uniform characteristics identical to the parent plant. Additionally, this technique can be applied for specific purposes beyond mass propagation. For example, another study [5] highlighted that in vitro culture plays an essential role not only in large-scale propagation of food crops but also in germplasm conservation through slow-growth storage and cryopreservation, and in plant breeding by supporting callus induction, somatic embryogenesis, and genetic transformation. Their study further emphasized that the success of these applications largely depends on the use of plant growth regulators (PGRs), particularly cytokinins and auxins, although there is growing interest in exploring natural alternatives as more sustainable options.
Banana plants can be efficiently propagated through micropropagation via callus formation or somatic embryogenesis [6], either directly or indirectly. Indirect embryogenesis occurs through the induction of callus formation (embryogenic callus), whereas direct embryogenesis bypasses the callus formation stage. The callus and/or embryogenic callus that forms then undergoes proliferation. The embryogenic callus subsequently enters the regeneration process, which consists of embryo maturation and germination of the mature embryo (cotyledon stage). Eventually, the embryo develops into a complete plantlet with shoots and roots [7]. Following this stage, the plantlet undergoes an enlargement process before acclimatization. Each stage of this process requires a basal culture medium supplemented with one or more plant growth regulators (PGRs), either as single agents or in combination [8, 9].
The plant growth regulators (PGRs) commonly used in these processes belong to the cytokinin, e.g., 6-benzylaminopurine (BAP) [10-12], auxin, abscisic acid (ABA), and gibberellin groups. PGRs function according to their specific roles when applied at the appropriate concentration. Cytokinin are primally required for shoot formation, and auxins for root development [13], ABA for maturation, and gibberellins for germination. If the cytokinin and auxin concentrations are balanced internally or externally, a callus or embryogenic callus will form [14], later developing into an embryo. The induction and proliferation of callus or somatic embryos can be achieved using a combination of BAP and 2,4-dichlorophenoxy acetic acid (2,4-D) (an auxin) [12].
Micropropagation media can also incorporate natural plant growth regulators (PGR) sources. Natural PGR sources commonly used in plant tissue culture as sources of cytokinin and auxin include coconut water, potato extract, carrot extract, shallot extract, and ripe banana fruit. Natural materials that have not yet been widely utilized for plant tissue culture but have undergone preliminary research include Aloe vera gel [15] and Vigna radiata sprout extract [16].
Although natural PGR sources such as coconut water and other fruit or tuber extracts have long been applied in tissue culture, the application of Aloe vera gel and Vigna radiata sprout extract remains underexplored and limited to preliminary reports. Plant-based extracts have been increasingly recognized as alternative sources of natural growth regulators in tissue culture systems. This study, therefore, provides one of the first empirical evaluations of these locally available, eco-friendly materials as natural PGR supplements in banana micropropagation, highlighting both their novelty and potential contribution to reducing dependence on synthetic PGRs.
Several studies have been conducted to analyze the bioactive compounds present in Aloe vera [17-21] and Vigna radiata sprouts [16, 22-24], similar to coconut water [25] and other natural materials. The compounds contained in Aloe vera gel juice and Vigna radiata sprout extract are expected to influence the growth direction and type of plant tissue culture in the micropropagation process of Gapi banana plants. Aloe vera gel juice and Vigna radiata sprout extract are hypothesized to induce shoot formation, callus proliferation, or somatic embryo development from Gapi banana explants, forming fully developed plantlets.
This study aims to evaluate the effects of adding Aloe vera gel juice and Vigna radiata sprout extract, compared to the control media (MS-0), BAP, and the combination of BAP with 2,4-D in Murashige and Skoog (MS) culture media for the micropropagation of Gapi banana plants.
2. MATERIALS AND METHODS
2.1. Research Place and Time
The research was conducted at the Plant Tissue Culture Laboratory, Faculty of Agriculture, Universitas Negeri Gorontalo, Province of Gorontalo, Indonesia, from July to December 2024.
2.2. Plant and Treatment Materials
The primary materials used in this study included Gapi banana suckers, Aloe vera gel juice, Vigna radiata sprout extract, BAP, and 2,4-D plant growth regulators (PGRs). Due to the limited availability of Gapi banana suckers as plant material for explants, a total of 26 explants (banana suckers) were used in this study, with two replications. This number excluded any explants that failed during the initial establishment phase.
2.3. Research Experimental Design and Treatments Media
The experiment was conducted under laboratory conditions using a Completely Randomized Design (CRD) consisting of 13 treatment combinations. The treatments were formulated using Murashige and Skoog (MS) basal media supplemented with Aloe vera gel juice (MS-LB) and Vigna radiata sprout extract (MS-ET) as natural plant growth regulator sources. These treatments were compared to control media, including MS without additives (MS-0), MS supplemented with 1 mg L-1 BAP (MS-BAP), and MS containing a combination of BAP (1 mg L-1) and 2,4-D at concentrations of 0.1, 0.3, and 0.5 1 mg L-1 (MS-BAP-2,4-D).
In the MS-LB treatments, the medium was supplemented with Aloe vera gel juice at concentrations of 10,15, 20, and 25% (v/v), while the MS-ET treatments contained Vigna radiata sprout extract at concentrations of 10, 15, 20, and 25% (v/v). Each treatment was replicated twice due to the limited number of available explants (banana suckers). Data collection was carried out through qualitative observations, including contamination occurrence, degree of browning, and regeneration responses such as shoot and callus formation.
2.4. Preparation of Control and Treatment Media
The control and treatment media were prepared using Murashige and Skoog (MS) basal medium. The composition of the MS basal medium per liter included MS mineral nutrients (from Himedia) 4.43 g, sucrose 3% (w/v), myo-inositol 10 mg L-1, and distilled water up to 1000 mL, with the pH adjusted to 5.8.
Aloe vera gel juice was obtained through the following procedure: the gel was separated from the skin and soaked in water overnight. It was then rinsed up to five times, drained, and blended until smooth. It was then passed through a coarse sieve to remove fibrous residua. The resulting gel juice was considered a crude extract and was directly used as a supplement in the Murashige and Skoog (MS) medium without further purification. Vigna radiata sprouts purchased from a local supplier were boiled with distilled water in a 1:1 ratio (w/v) until boiling. The resulting boiling water extract was collected and used directly as a crude supplement in MS medium without further purification. The filtration of Vigna radiata sprout extract was performed solely to separate the liquid fraction from the sprout residues.
Aloe vera gel juice was added to the MS medium for the MS-LB treatment media at concentrations of 10, 15, 20, and 25% (v/v). Vigna radiata sprout extract was added to the MS medium for the MS-ET treatment media at concentrations of 10, 15, 20, and 25% (v/v).
As a comparison, MS-0 medium was used as the control, along with MS medium supplemented with BAP 1 mg L-1 and a combination of BAP 1 mg L-1 with 2,4-D at concentrations of 0.1 and 0.3. and 0.5 mg L-1. Finally, 5 g L-1 of micropropagation agar was added. After thoroughly mixing all the ingredients, the solution was heated to dissolve the agar. The prepared medium was then distributed into culture bottles and sterilized using an autoclave at 121°C under 115 kPa pressure for 20 minutes.
2.5. Initiation and Sterilization of Explants
The explants were obtained from Gapi banana suckers, cleaned of any adhering soil, and the corm (pseudostem) was removed. The explants were then washed with a detergent solution while being brushed and rinsed with water until completely clean. The rhizome section was trimmed to approximately an adult's thumb size.
The sterilization process was conducted inside a laminar airflow cabinet (LAF) using a 20% (v/v) disinfectant solution for 20 minutes, followed by a 10% (v/v) disinfectant solution for 15 minutes. After each sterilization treatment, the explants were rinsed three times with sterile distilled water. The sterilized explants were then trimmed to a size of approximately 1 cm2, forming a small stupa-like structure. The explants were then divided into two equal parts.
2.6. Inoculation and Incubation of Explants
After preparation, the Gapi banana explants were inoculated into the respective treatment media inside culture bottles. Due to the limited availability of explant sources, only two culture bottles were prepared for each treatment. Each bottle contains one explant. The culture bottles were then placed in an incubation room at a temperature of 25 ± 2°C, under LED light (9 watts), with a photoperiod of 16 hours of light and 8 hours of dark. Each month, subculturing was performed into fresh medium treatments.
2.7. Observations and Data Analysis
All data collected from culture observations were analyzed both qualitatively and statistically. Observations were recorded at one-month intervals, focused primarily on the percentage of contamination, the percentage of browning, and explant growth responses, specifically whether shoots/roots, calluses were formed in the 1st, 2nd, and 3rd months.
2.7.1. Contamination
The percentage of contamination was calculated to evaluate the frequency of microbial infection during culture initiation. Contamination (%) was determined using the following formula:
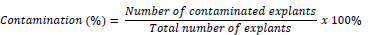
The calculation clarifies how values such as 0% contamination were obtained in the experimental results.
2.7.2. Browning
Browning was recorded as the progressive darkening of Gapi banana explants, beginning at the cut surfaces, and caused by phenolic oxidation. In this study, browning was defined qualitatively based on the intensity and spread of discoloration: mild browning (localized at the cut edge) indicated viability, whereas extensive browning across most tissue indicated severe damage. Observations were compared across Aloe vera, Vigna radiata, and synthetic PGR treatments to assess their effects on reducing oxidative stress and maintaining explant viability. Browning (%) was determined using the following formula:
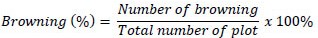
2.7.3. Explain Growth Responses
The observations focused on comparing the development of Gapi banana explants under different culture media treatments. Specifically, data were collected on whether shoots, roots, or callus tissue were formed during the first, second, and third months of culture. Observations were carried out visually at monthly intervals, supported by photographic documentation. This approach enabled a comparative assessment of explant responses to Aloe vera gel juice, Vigna radiata sprout extract, and synthetic PGRs, highlighting differences in their ability to support growth and regeneration under in vitro conditions.
The categorical data were then converted into binary form, where a value of 1 represented active growth and 0 represented no growth. The Chi-square (χ2) test was applied to determine whether significant differences existed among treatments in terms of explant survival and growth response during the first, second, and third months of culture. Statistical significance was evaluated at the 5% probability level (p < 0.05). Data were analyzed using STAR software version 2.0.1 (Biometrics and Breeding Informatics, IRRI, Los Banos, Philippines).
3. RESULTS AND DISCUSSION
3.1. Results
3.1.1. Effect of Culture Media on Explant Survival
During the first month of culture, all treatments exhibited browning symptoms (100%), as shown in Table 1. Browning began at the cut surfaces and gradually spread throughout the explant tissues during culture.
Contamination was observed only in the MS-ET 10% medium (100%), while all other treatments remained sterile (0%), indicating that the sterilization procedure was generally effective. Despite the contamination, explants cultured in MS-ET 10% produced early shoot growth within the first month after inoculation.
Explants grown on MS-LB (Aloe vera gel juice) media at concentrations of 10-25% and most MS-ET treatments (15-25%) showed complete blackening and no visible growth. Explants in the control medium (MS-0) also failed to develop.
The Chi-square (χ2) test revealed significant differences among treatments in explant survival (χ2 = 26, df = 12, p = 0.0107) (Table 2). This statistical result confirmed that the type of culture medium had a significant effect on explant response during the first month of culture.
| S.No. | Culture Media Treatment | Percentage of Contamination (%) | Browning (%) | Inoculant Condition in the First Month |
|---|---|---|---|---|
| 1. | MS-0 (Control) | 0 | 100 | Not growing |
| 2. | MS-LB 10% | 0 | 100 | Blackened |
| 3. | MS-LB 15% | 0 | 100 | Blackened |
| 4. | MS-LB 20% | 0 | 100 | Blackened |
| 5. | MS-LB 25% | 0 | 100 | Blackened |
| 6. | MS-ET 10% | 100 | 100 | Shoots grow |
| 7. | MS-ET 15% | 0 | 100 | Blackened |
| 8. | MS-ET 20% | 0 | 100 | Blackened |
| 9. | MS-ET 25% | 0 | 100 | Blackened |
| 10. | MS-BAP 1 mg.L-1 | 0 | 100 | The inoculant enlarges |
| 11. | MS-BAP 1 mg.L-1 - 2,4 D 0.1 mg.L-1 | 0 | 100 | The inoculant enlarges |
| 12. | MS-BAP 1 mg.L-1 - 2,4 D 0.3 mg.L-1 | 0 | 100 | The inoculant enlarges |
| 13. | MS-BAP 1 mg.L-1 - 2,4 D 0.5 mg.L-1 | 0 | 100 | Blackened |
| S.No. | Culture Media Treatment |
Alive (n) (growing) |
Dead (n) (blackened/not growing) |
Expected Alive | Expected Dead |
|---|---|---|---|---|---|
| 1. | MS-0 (Control) | 0 | 2 | 0.62 | 1.38 |
| 2. | MS-LB 10% | 0 | 2 | 0.62 | 1.38 |
| 3. | MS-LB 15% | 0 | 2 | 0.62 | 1.38 |
| 4. | MS-LB 20% | 0 | 2 | 0.62 | 1.38 |
| 5. | MS-LB 25% | 0 | 2 | 0.62 | 1.38 |
| 6. | MS-ET 10% | 2 | 0 | 0.62 | 1.38 |
| 7. | MS-ET 15% | 0 | 2 | 0.62 | 1.38 |
| 8. | MS-ET 20% | 0 | 2 | 0.62 | 1.38 |
| 9. | MS-ET 25% | 0 | 2 | 0.62 | 1.38 |
| 10. | MS-BAP 1 mg L-1 | 2 | 0 | 0.62 | 1.38 |
| 11. | MS-BAP 1 mg L-1 - 2,4 D 0.1 mg L-1 | 2 | 0 | 0.62 | 1.38 |
| 12. | MS-BAP 1 mg L-1 - 2,4 D 0.3 mg L-1 | 2 | 0 | 0.62 | 1.38 |
| 13. | MS-BAP 1 mg L-1 - 2,4 D 0.5 mg L-1 | 0 | 2 | 0.62 | 1.38 |
| Total | 8 | 18 | |||
| Chi-square (χ2) = 26.00 | Df = 12 | P-value = 0.0107 (<0.05) |
The MS-ET 10% medium and the MS-BAP-based treatments showed the highest survival rates, with 100% of explants exhibiting enlargement or shoot formation. In contrast, explants in the MS-LB (Aloe vera gel juice) and control (MS-0) media showed no growth.
3.1.2. Culture Condition in The Micropropagation of Gapi Banana (1st, 2nd, and 3rd Month)
The culture responses of Gapi banana explants differed among treatments during the three months of observation (Table 3). In the control (MS-0) medium, explants remained dormant with no visible growth throughout the culture period (Fig. 1).
In the MS-LB treatment (10 to 25% (v/v)) (Fig. 2), the explant initially blackened during the first month but began to exhibit growth in the second and third months in some treatments. MS-LB 10% and 15% (v/v) started to show either shoot or root growth in the second month, which later developed into shoots in the third month. The MS-LB 20% (v/v) treatment exhibited the best growth, with shoot formation in the second month and the development of a complete plantlet in the third month. Conversely, MS-LB 25% (v/v) failed to develop throughout the three months, indicating that a higher concentration (25% v/v) of Aloe vera gel juice did not optimally support the growth of Gapi banana explants.
| S.No. | Culture Media Treatment | Culture Condition in the 1st Month | Culture Condition in the 2nd Month | Culture Condition in the 3rd Month |
|---|---|---|---|---|
| 1. | MS-0 (Control) | Not growing | Not growing | Not growing |
| 2. | MS-LB 10% | Blackened | Shoots grow | Potential shoots |
| 3. | MS-LB 15% | Blackened | Roots grow and appear green at one end of the inoculant | shoots |
| 4. | MS-LB 20% | Blackened | Shoots grow | Plantlets |
| 5. | MS-LB 25% | Blackened | Not growing | Not growing |
| 6. | MS-ET 10% | Shoots grow | Shoots grow | Shoots grow bigger |
| 7. | MS-ET 15% | Blackened | Not growing Stay black |
Not growing Stay black |
| 8. | MS-ET 20% | Blackened | Looks like going to sprout | Shoots grow |
| 9. | MS-ET 25% | Blackened | Stay black | Stay black |
| 10. | MS-BAP 1 mg L-1 | The inoculant enlarges | Shoots and callus grow | Several shoots are formed |
| 11. | MS-BAP 1 mg L-1 - 2,4 D 0.1 mg L-1 | The inoculant enlarges | Forms a compact callus | compact callus |
| 12. | MS-BAP 1 mg L-1 - 2,4 D 0.3 mg L-1 | The inoculant enlarges | Forms a compact callus | compact callus |
| 13. | MS-BAP 1 mg L - 2,4 D 0.5 mg L-1 | Blackened | Stay black | Stay black |

Condition of Gapi banana explants in the MS-0 (control) medium from the first month (1), second month (2), to the third month (3).

Condition of explants in the MS-LB (Aloe vera gel juice) treatment medium, concentrations: (a) 10%, (b) 15%, (c) 20%, and (d) 25%, observed from the first month, the second month, and the third month.

Condition of explants in the MS-ET (Vigna radiata sprout extract) treatment medium, concentrations: (a) 10%, (b) 15%, (c) 20%, and (d) 25%, from the first month, the second month, to the third month.
The MS medium supplemented with Vigna radiata sprout extract (MS-ET) exhibited variable results. The MS-ET 10% (v/v) treatment proved to be the most effective in stimulating stable shoot growth, with shoots continuing to enlarge until the third month. The MS-ET 20% (v/v) treatment also showed positive explant growth, as the explant began turning green in the second month and developed shoots in the third month (Fig. 3). In contrast, the explants in the MS-ET 15 and 25% (v/v) treatments remained blackened and showed no growth or development. This indicates that the formulation of these media concentrations was unsuitable for explant regeneration.
In the first month, the explants exhibited size enlargement in the MS-BAP 1 mg L-1 treatment and in the combination treatments of MS-BAP 1 mg L-1 + 2,4-D at 0.1 and 0.3 mg L-1. In the second month, explants in the MS-BAP 1 mg L-1 treatment and the MS-BAP 1 mg L-1 + 2,4-D (0.1 and 0.3 mg L-1) combinations began to form shoots and compact callus. Meanwhile, the explants in the MS-BAP 1 mg L-1 + 2,4-D 0.5 mg L-1 treatment showed no growth and remained blackened. This indicates an inhibitory effect at this concentration (Fig. 4).
In the third month, the explant in the MS-BAP 1 mg L-1 treatment successfully developed multiple shoots, while the combinations of MS-BAP 1 mg L-1 + 2,4-D at 0.1 and 0.3 mg L-1 remained as compact callus. This indicates that these treatment media have a high potential for inducing callus formation, which may later develop into shoots. However, in the MS-BAP 1 mg L-1 + 2,4-D 0.5 mg L-1 treatment, the explant remained blackened up to the third month, suggesting that an increased concentration of 2,4-D in this medium may have hurt explant viability (Fig 4).

Condition of Gapi banana explants in the MS-BAP and BAP-2,4-D combination media, concentrations: (a) BAP 1 mg L-1, (b) BAP 1 mg L-1 + 2,4-D 0.1 mg L-1, (c) BAP 1 mg L-1 + 2,4-D 0.3 mg L-1, (d) and BAP 1 mg L-1 + 2,4-D 0.5 mg L-1, observed from the first month (1), second month (2), to the third month (3).
Overall, differential growth responses were recorded, with explants showing active development mainly in MS-LB 20%, MS-ET 10%, and MS-BAP-based media (Table 3). These findings formed the basis for the subsequent statistical evaluation using Chi-square analysis.
3.1.3. Chi-square Analysis of Growth Response
The Chi-square (χ2) test showed significant differences among treatments during the first month (χ2 = 26.00, df = 12, p = 0.0107), indicating that the culture medium composition significantly affected the early growth response of Gapi banana explants. During the second (χ2 = 13.00, df = 12, p = 0.369) and the third month (χ2 = 13.00, df = 12, p = 0.369), no significant differences were observed among treatments (p > 0.05), suggesting that once the explants had initiated growth, their subsequent developmental pattern remained stable across treatments (Table 4).
| Observation Month | χ2Value | df | p-value | Significance |
|---|---|---|---|---|
| Second month | 13.00 | 12 | 0.369 | ns (p>0.05) |
| Third month | 13.00 | 12 | 0.369 | ns (p>0.05) |
4. DISCUSSION
4.1. The Effect of Aloe vera Gel Juice on the Micropropagation Development of Gapi Banana
The research results on the MS-LB treatment medium, which consists of MS medium supplemented with Aloe vera gel juice, showed that the explant initially blackened, resembling cell death symptoms. However, over time, it recovered and began to develop shoots. This phenomenon can be explained by the bioactive compounds in Aloe vera gel, which play a crucial role in supporting cell regeneration and the growth of plant tissue.
Aloe vera gel contains polysaccharides, enzymes, vitamins, minerals [26], and natural plant growth hormones such as auxins and gibberellins. These growth hormones are known to stimulate cell division and tissue regeneration. Additionally, polysaccharides in Aloe vera exhibit antioxidant properties, which help reduce oxidative stress caused by the accumulation of phenolic compounds [27]. Phenolic compounds are the primary cause of browning and tissue death in plant tissue culture, making the antioxidant function of Aloe vera particularly beneficial for enhancing the survival and development of explants.
Additionally, the amino acids and phenolic compounds present in Aloe vera gel can act as cell protectors against toxic compounds while accelerating the recovery of damaged cells [21]. Therefore, although the explant initially blackened due to tissue culture stress, the presence of Aloe vera gel juice in the medium helped in tissue recovery [28] and stimulated shoot growth, demonstrating its potential as a natural supplement to enhance the success of Gapi banana micropropagation. This finding also highlights Aloe vera gel as a possible solution to the challenge of obtaining Gapi banana explants for in vitro culture, providing an alternative approach to improve explant viability and regeneration [26]. A previous study [29] showed that Aloe vera gel coating can extend the shelf life of lime by up to 4 days by slowing down colour changes without affecting the physicochemical quality of the fruit.
4.2. The Effect of Vigna Radiata Sprout Extract on the Culture Development of Gapi Banana
The research results on the MS-ET treatment medium, which consists of MS medium supplemented with Vigna radiata sprout extract, showed varied growth responses among the explants. Some explants turned green and developed shoots, while others remained blackened without further growth. Vigna radiata sprout extract is rich in natural plant growth hormones, particularly cytokinin. Germinated Vigna radiata sprouts accumulate bioactive metabolites and exhibit cytokinin-related biological activity, supporting their role as natural PGR sources [30]. Cytokinin is crucial in stimulating cell division and organogenesis in tissue culture. They also function to delay tissue culture senescence (aging) and accelerate shoot formation by inducing the expression of genes associated with cell division and differentiation [31]. Additionally, Vigna radiata sprout extract contains proteins, essential amino acids, and antioxidant enzymes [32-34], which can enhance explant viability and reduce the impact of stress caused by in vitro culture. However, this study observed that not all explants in the MS-ET medium successfully developed shoots.
This could be due to the suboptimal concentration of sprout extract or the presence of certain compounds that may cause an imbalance in explant growth. Therefore, optimizing the Vigna radiata sprout extract concentration in the culture medium is necessary to maximize the positive effects of cytokinin and other bioactive components, thereby improving shoot regeneration in the micropropagation of Gapi banana plants. According to [35], mung beans contain flavonoids, polysaccharides, and peptides, which act as antioxidants [36], neutralizing free radicals, reducing inflammation, and preventing chronic diseases such as diabetes and cancer [37].
4.3. The Effect of Benzylaminopurine (BAP) and 2,4-Dichlorophenoxy acid (2,4-D) on the Culture Development of Gapi Banana
The research results showed that the addition of BAP and 2,4-D in the culture medium had a significant influence on the development of Gapi banana explants. In the treatment with MS medium + BAP 1 mg L-1, the explants initially enlarged, then began to form shoots and callus, eventually producing several small shoots. This indicates that BAP, as a cytokinin, plays a role in stimulating cell division and shoot differentiation [38, 39]. Cytokinins are known to regulate plant growth by promoting shoot formation and accelerating cell division in the meristematic region.
However, when BAP was combined with 2,4-D at different concentrations, the explant responses varied significantly. In the MS medium containing BAP 1 mg L-1 + 2,4-D 0.1 mg L-1, the explants enlarged and formed compact callus, but no shoot formation was observed. A similar phenomenon occurred in the treatment with BAP 1 mg L-1 + 2,4-D 0.3 mg L-1, where the explants continued to form compact callus without any shoot differentiation. This can be explained by the dominant effect of auxin (2,4-D) in inducing callus formation [40], while cytokinin at the same concentration was not strong enough to stimulate shoot formation. Auxin plays a key role in callus induction and tissue regeneration initiation, but when its concentration is higher than that of cytokinin, shoot formation may be inhibited [39].
In the treatment with BAP 1 mg L-1 + 2,4-D 0.5 mg L-1, the explants turned black and remained black until the end of the observation period, indicating necrosis or tissue death. In this study, necrosis was defined as intense blackening in more than 75% of explant tissue that persisted until the end of the observation period. The high concentration of 2,4-D likely caused physiological stress on the explants, disrupting cellular metabolism and inhibiting growth. Additionally, an increase in auxin levels in the medium is known to trigger phenolic accumulation, leading to browning and tissue death in in vitro cultures [38].
Thus, the combination of BAP and 2,4-D must be optimized to promote shoot proliferation without causing excessive callus formation or necrosis [40]. Based on these findings, BAP without 2,4-D was more effective in inducing shoot growth, whereas low concentrations of 2,4-D were more suitable for callus induction. Meanwhile, a high concentration of 2,4-D is not recommended, as it can damage plant tissues [39]. According to the previous studies [38, 41], the presence of cytokinin, along with a small amount of auxin, is required for haploid tissue to form callus and somatic embryos. Understanding the interaction between these two hormones is essential for developing a more optimal tissue culture technique for Gapi banana, whether for shoot multiplication or callus production in further studies.
Overall, the findings suggest that Aloe vera gel juice and Vigna radiata sprout extract have the potential for the micropropagation of Gapi banana. These natural supplements demonstrated comparable to, or even superior to, conventional synthetic PGRs, thereby emphasizing their practical value in plant tissue culture. While the current study was limited to controlled laboratory conditions, the consistency of the results provides a solid basis for considering broader applications. Building upon these findings, it is essential to reflect on the potential prospects and future directions that may further enhance the applicability of plant-based PGRs in banana micropropagation and beyond.
The findings of this study highlight the practical potential of Aloe vera gel juice and Vigna radiata sprout extract as natural plant growth regulators (PGR) sources in banana micropropagation. Beyond the current laboratory-based observations, these results open prospects for applying locally available plant extracts as low-cost and eco-friendly supplements in tissue culture media. Further studies are recommended to validate these findings under larger sample sizes, explore concentration optimization, and assess reproducibility in field-scale trials. Such efforts will strengthen the potential for large-scale banana propagation and contribute to the development and commercialization of bio-based PGRs.
5. STRENGTHS AND LIMITATIONS
One of the key strengths of this study was the utilization of natural, locally available materials as PGR alternatives.
This study was limited by the relatively small number of explants used due to the restricted availability of Gapi banana suckers as planting material.
Additionally, browning of explants was observed in all treatments, which may have affected regeneration performance. The study was conducted under controlled laboratory conditions, and therefore, the results may not fully reflect responses under large-scale or field conditions. Future research should include larger sample sizes, explore optimization of Aloe vera and Vigna radiata concentrations, and test the reproducibility of results under different environmental and field conditions.
CONCLUSION
This study demonstrated that the type and composition of culture media significantly affected the in vitro response of Gapi banana explants. MS medium supplemented with Aloe vera gel juice (MS-LB 20%, v/v) and Vigna radiata sprout extract (MS-ET 10%, v/v) was the most effective in inducing shoot formation, outperforming the synthetic cytokinin BAP during the same culture period. The combination of BAP (1 mg L-1) with low concentrations of 2,4-D (0.1-0.3 mg L-1) promoted compact callus formation, whereas higher 2,4-D levels (0.5 mg L-1) and high Aloe vera or sprout extract concentrations inhibited growth due to tissue blackening. Overall, the results highlight the potential of using Aloe vera gel juice and Vigna radiata sprout extract as eco-friendly and low-cost alternatives to synthetic PGRs for banana micropropagation, providing a basis for further optimization and scale-up studies.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contributions to the paper as follows: I.H. and J.A.: Study conception and design were carried out; I.H., J.A., and D.B.P.: Data collection was performed; I.H., R.S., and H.D. Analysis and interpretation of results were conducted; I.H., D.B.P., and H.D.: Drafting of the manuscript was done. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| MS | = Murashige and Skoog medium |
| MS-0 | = MS medium without supplements (control) |
| MS-LB | = MS medium + Aloe vera gel juice |
| MS-ET | = MS medium + Vigna radiata sprout extract |
| MS-BAP | = MS medium + 6-Benzylaminopurine |
| MS-BAP-2,4-D | = MS medium + BAP and 2,4-Dichlorophenoxyacetic acid |
| PGR | = Plant Growth Regulator |
| CRD | = Completely Randomized Design |
AVAILABILITY OF DATA AND MATERIALS
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request. All materials used in this study are described in the Methods section to ensure reproducibility.
FUNDING
This research was financially supported by the Ministry of Education, Culture, Research, and Technology of the Republic of Indonesia (Kemendikbudristek) through Universitas Negeri Gorontalo (State University of Gorontalo) through Penelitian Fundamental-Reguler (PRF) 2024 No.: 459/E5/PG.02.00/2024 (2024/05/30) under Contract No.: 063/E5/PG.02.00.PL/2024 (2024/06/11), via Decree of Rector of Universitas Negeri Gorontalo No.: 733/UN47/HK.02/2024 (2024/11/12).
ACKNOWLEDGEMENTS
The authors would like to thank Dean and the staff of the Plant Tissue Culture Laboratory, Faculty of Agriculture, Universitas Negeri Gorontalo (State University of Gorontalo), for their technical assistance during the experiment. The authors are also grateful to the Research and Community Service Institute (LPPM) of Universitas Negeri Gorontalo (UNG) for administrative support.


