All published articles of this journal are available on ScienceDirect.
Evaluation of Fixed-Bed Cultures with Immobilized Lactococcus Lactis ssp. Lactis on Different Scales
Abstract
Fixed-bed processes, where cells are immobilized within macroporous carriers, are a promising alternative to processes with suspended cells. A scale-up concept is presented in order to evaluate the performance as part of process design of fixed-bed processes. Therefore, Lactococcus lactis cultivation in chemostat and batch mode was compared to fixed bed cultures on three different scales, the smallest being the downscaled Multiferm with 10 mL fixed bed units, the second a 100 mL fixed-bed reactor and the third a pilot scale reactor with 1 L fixed bed volume. As expected, the volume specific lactate productivity of all cultivations was dependent on dilution rate. In suspension chemostat culture a maximum of 2.3 g·L-1·h-1 was reached. Due to cell retention in the fixed-beds, productivity increased up to 8.29 g·L-1·h-1 at a dilution rate of D = 1.16 h-1 (corresponding to 2.4·µmax) on pilot scale. For all fixed bed cultures a common spline was obtained indicating a good scale-up performance.
INTRODUCTION
Lactic acid bacteria (LAB) are commonly used in the production of fermented dairy products as well as for production of lactic acid, antimicrobial substances (bacteriocins) and biodegradable polymers, among others [1-3]. Industrial processes use mostly conventional batch or fed-batch fermentation with suspended cells. Reactor volumes go up to 100 m3, process time varies between several hours and days depending on the strain and the process strategy [2]. Even if high cell and product concentrations can be reached, the known drawbacks such as low productivity, product inhibition and also the variation from batch to batch remain [4-6].
Continuous cultivation of suspended cells is mainly performed with cell-recycle in perfusion mode to enable high cell concentration and product productivity [7]. For cell-recycle often filtration units have been applied (hollow fiber modules, ceramic membrane modules etc., reviewed in [7]). When used in long-term experiments, however, it is usually difficult to avoid membrane fouling due to cell growth. Furthermore, scale-up to industrially relevant scales has not been solved sufficiently yet.
Immobilization of LAB has many advantages compared to cell suspension culture such as high volumetric productivity, improved production efficiency and no possibility of washout [8-11]. Fixed-bed processes, where cells are immobilized within macroporous carriers, are very promising in this respect [7, 10-13]. Immobilization of cells on inert porous carriers is an easy way to retain biomass [13-16]. Due to cell retention it is possible to run fixed-bed bioreactors in a perfusion mode at a steady-state with dilution rates higher than the maximum specific growth rate of the used strain. By this, very high volume specific productivity with respect to lactate can be reached and maintained for long periods of time. We recently reported on fixed-bed cultures of L. lactis, where the volume-specific lactic acid production rate was significantly higher as in batch culture, continuous cultivation could be maintained for approx. 50 days, and a successful scale-up to 1 L-scale with perfusion rates of up to approx. 35 L per day [7].
Nevertheless, the number of industrial fixed-bed processes is quite small. To some extent this is due to the lack of process development tools for fixed-bed processes. To fill this gap, a strategy was developed for the design and evaluation of relevant process parameters of fixed-bed processes (Fig. 1). The first, very small scale of 10 mL working volume is the multi-fixed-bed bioreactor “Multiferm”. We recently reported on the successful application of this system for the first evaluation of process parameters and kinetic modelling [17]. The next step is a 100 mL fixed-bed system, which can be operated continuously with reasonable effort to investigate the performance and long-term-stability of the culture. For small fixed-bed volumes with a height of approximately 10 cm, the medium can be pumped axially through the bed. In this case the pH at the outlet should remain in a physiological range. A further increase of the length would result in too low pH-values in the upper zones of the bed. This can be overcome by applying a radial medium flow, where the radius determines the length of the pH gradient, not the height of the column [7, 18]. As the first approach for scale-up, a 1 L fixed-bed reactor is applied. Even if this is probably not the final industrial scale, the reactor system already has the main characteristics of a large-scale system, mainly the radius. For further increase of the volume, just the height has to be increased [19].

For all the three systems, a “proof-of-concept” has been shown before [7, 17]. The aim of the study presented here was to compare the performance of these three fixed-bed systems with Lactococcus lactis ssp. lactis regarding lactic acid production, biomass formation and lactose consumption. Furthermore, batch and chemostat cultures with suspended cells were performed to evaluate the productivity of the fixed-bed system.
In earlier studies of our group [7] the carrier CERAMTEC EO/90 gave promising results with respect to cell activity and long-term stability. But due to the form of the carrier (ring with inner diameter of 3 mm, outer diameter of 8 mm, height 8 mm) the structure of the bed was non-homogeneous. Cells could not grow within the carriers due to the very low pore size (approx. 2.5 µm). Therefore, the risk for wash-out of cells was very high. Furthermore, the carrier had not been developed and optimized for biological applications. As an alternative a new, macroporous glass carrier (VitraPOR®, ROBU® Glasfilter-Geräte GmbH), which had been developed especially for cell cultivation, was used here.
MATERIALS AND METHODS
Bacterial Strain
For this study a Lactococcus lactis ssp. lactis strain (DSM-No. 20481 type strain, abbreviated as L. lactis) was used. It is a homofermentative strain that produces L-lactate under mesophilic growth conditions (30 °C, pH 7). Like many other lactic acid bacteria, it is anaerobic but aerotolerant. As standard medium M17 broth (DifcoTM, BD) was used, which was supplemented with lactose to a final concentration of 5 g·L-1. Medium and lactose solutions were autoclaved separately to avoid loss of substrate and nutrients due to Maillard’s reaction.
Suspension Culture
Inoculum Preparation
For each experiment a fresh pre-culture was prepared from a cryostock as described previously [20]. This pre-culture was used as inoculum for experiments in the different reactors.
Bioreactor Set-up for Batch Cultivation of Suspended Cells
Batch cultivations with pH control were performed in the bioreactor system Vario 500 (medorex e.K., Germany), with a maximum working volume of 450 mL. The double walled jacket was connected to a separate circulating water bath for temperature control. The axial propeller stirrer was driven at 300 rpm. The pH was controlled at 7.0 ± 0.1 by feeding of 5 M NaOH aq through a peristaltic pump coupled to an integrated pH electrode. Subsequently, the whole system was filled with 300 mL dH2O and autoclaved for 15 min at 121 °C.
Before inoculating the fermenter with 1·108 cells∙mL-1, the dH2O was replaced by 360 mL medium and the bioreactor system was installed and ran overnight in order to let the medium reach the set temperature of 30 °C and to assess sterility. The next morning, a total of 4·1010 cells from pre-culture were prepared in 20 mL fresh medium and inoculated with a Luer lock syringe. Subsequently, 20 mL of medium were then added to achieve the working volume of 400 mL and to wash cells from the inoculation pipe. Samples of 12 mL were taken every hour and assessed for biomass, lactate and lactose. Fermentations were performed three times.
Maximum specific growth rate was determined from the slope of the linear regression line of logarithmic plotted mean biomass data of these batch fermentations. This slope gave µmax = 0.49 h-1. Equations are given in [20].
Bioreactor Set-up for Continuous Chemostat Cultivation of Suspended Cells
Chemostat experiments were carried out in the multi-fermentation system DASGIP® (Eppendorf AG, Germany). Experiments were performed under pH and temperature control to assess optimal growth conditions. Three reactor units with a working volume of 350 mL were run independently and in parallel. Each unit had an own medium reservoir from where medium at room temperature was fed through peristaltic pumps. Samples were withdrawn regularly for analyses of biomass, lactate and lactose courses. The dilution rates were chosen as 0.2, 0.4, 0.6, 0.8 and 0.9 times the maximum specific growth rate. For further details see [20].
Fixed-bed Cultivation
Cultivations of immobilized bacteria were carried out in fixed-bed bioreactors of different scales (Fig. 1), beginning with the multiple fixed-bed bioreactor system Multiferm, then in a 100 mL-fixed-bed bioreactor and at last in a 1 L-fixed-bed bioreactor (all from medorex e. K, Nörten-Hardenberg, Germany).
Multiferm Fixed-bed System
The Multiferm comprises twelve single fixed-bed units consisting of a glass vessel, which is packed with the carrier material (10 mL, Fig. (1)). This bioreactor system was established and precisely described previously [17, 20]. Each unit was filled with borosilicate carriers (VitraPOR®, ROBU® Glasfilter-Geräte GmbH) with a mean diameter of 4 mm. The pH was not controlled. Temperature was controlled at 30 °C. The applied dilution rate was 0.5 h-1. Samples were taken at the outlet (harvest flow) of each fixed-bed unit.
For inoculation of the fixed-beds, a volume of 15 mL medium containing 109 cells was transferred to every unit and incubated for 16 h overnight. The next morning the perfusion was started after a replacement of the exhausted medium. During continuous operation, samples were taken every 48 h.
100 mL-Fixed-bed Bioreactor
The 100 mL-fixed-bed bioreactor consists of two cylindrical glass vessels. The outer vessel (1 L) serves as heating chamber for the inner glass vessel, which is packed with the carrier material (100 mL, Fig. (1)). The principal set-up of the inner vessel is similar to the fixed-bed unit of the Multiferm (see above). For more details see [20]. Again, the borosilicate carrier (VitraPOR®, ROBU® Glasfilter-Geräte GmbH) with a mean diameter of 4 mm was used. The pH was not controlled. Temperature was controlled at 30 °C with a Pt100 sensor and a heating bath. A magnetic bar with a magnetic stirrer was used for mixing within the outer vessel. For continuous operation the fixed-bed bioreactor was connected to a storage tank with fresh medium and a harvest tank. Medium feed and harvest was done by peristaltic pumps. Samples were taken at the outlet (harvest flow).
For inoculation, 100 mL of a pre-culture were pumped into the empty bed. Cells settled within the fixed-bed during 16 h of incubation. Afterwards perfusion was started.
1 L-Fixed-bed Bioreactor
The 1 L-fixed-bed bioreactor consists of a 3.5 L double-walled glass vessel (Fig. 1). Into this vessel the wire-cage for the carrier bed (bed volume 1 L) is integrated. Within the carrier bed, the circulating medium flows from an inner distribution tube parallel to the radius (radial flow). The radius corresponds to the height of the axial-flow fixed-bed bioreactors introduced above. For more details see [19]. Here the borosilicate carrier (VitraPOR®, ROBU® Glasfilter-Geräte GmbH) with a mean diameter of 8 mm was used because the smaller size would fall through the mesh of the of the wire-cage. Additionally, the cultivations with the smaller carrier showed that channeling could occur after long cultivation periods due to strong cell growth. The pH was not controlled during continuous cultivation. Temperature was controlled at 30 °C with a Pt100 sensor and a heating bath. A magnetic bar with a magnetic stirrer was used for mixing within the vessel. For continuous operation the fixed-bed bioreactor was connected to a 355 L storage tank with fresh medium and a harvest tank. Medium feed and harvest was done by peristaltic pumps. Samples were taken at the outlet (harvest flow).
For inoculation 600 mL of a pre-culture were pumped into the empty bed. Cells settled within the fixed-bed during 18 h of incubation. After that perfusion was started.
Analytics
Biomass
Fermentation broth was analyzed on biomass content by measuring optical density at 600 nm (OD600). A previously determined correlation between OD and total cell concentration (TCC) was used to convert OD values to biomass content [20]:
| TCC=0D600 · 7.36 · 108 | (cells·mL-1) |
Lactate and Lactose Concentration
Substrate and product concentrations were determined by HPLC measurements with two devices as described previously [20] with the following parameters:
- Cation exchange column (pre-warmed to 40 °C), 2.5 mM sulfuric acid as eluent, 0.3 mL·min-1, refractive index detector for both components.
- Cation exchange column (pre-warmed to 50 °C), refractive index detector for lactose, UV-detector for lactate.
Cell Viability
Cell viability was determined by fluorescence staining with acridine orange (AO) / propidium iodide (PI). The cell suspension was diluted with 0.9% NaCl, mixed with the staining solution (v/v 1:1), incubated for 2 minutes and then centrifuged at 3·104·g for 2 minutes. The supernatant was discarded, the cell pellet was resuspended in 0.9% NaCl and centrifuged again. The supernatant was discarded, the cell pellet was resuspended in 0.9% NaCl. 10 µl of this solution was filled into a microscope slide. Under a fluorescence microscope dead cells (red) and viable cells (green) were counted.
RESULTS AND DISCUSSION
Relationship Between Maximal Growth Rate and pH
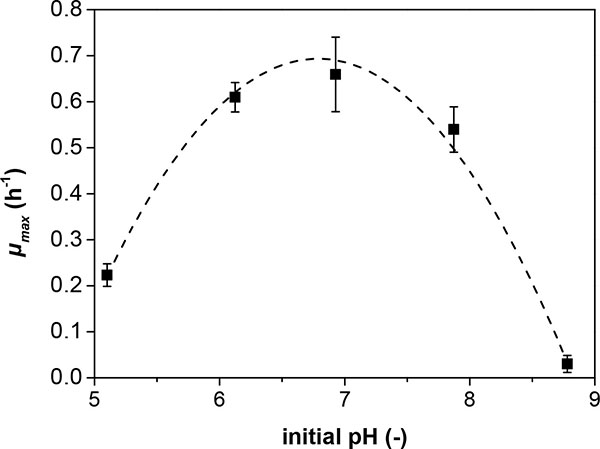
In fixed-bed bioreactors a pH-gradient along the length of the bed is inherent to the system [7]. The radial flow geometry has been suggested to account for this effect. Nevertheless it is important to judge, if the pH-values at the outlet of the bed is still tolerable or unfavorable for the cells. An experimental series was performed to determine the relationship between the medium pH and the maximum specific growth rate μmax in shaking flasks without pH control. Fig. (2) illustrates the determined μmax values at different initial pH values. The values for μmax were determined from logarithmic plots of OD600 data from the exponential growth phase of shaking flask experiments with the indicated initial pH values. The presented data points are the means ± SD of three independent experiments. The maximum μmax was scored at an initial pH of 6.9. Mild growth could still be observed at an initial medium pH as low as pH 5 and as high as 8.8, whilst no growth was observed at a pH of 4.0 (data not shown).
Fixed-bed Cultures on Different Scales
Results obtained with the Multiferm system were already presented previously [20]. The volume specific lactate productivity as an indicator of the cell activity in the fixed-bed is highlighted here, as it was used as parameter to compare different reactor systems. At a constant dilution rate of D = 0.5 h-1 the volume specific lactate productivity reached a mean steady-state value of 4.49 g·L-1·h-1. Standard deviation from parallel runs was below 5%, indicating a high reproducibility of the system.
Performance and long-term stability during a long-term cultivation in the 100 mL-fixed-bed bioreactor were investigated by increasing the dilution rate step-wise (Fig. 3). After incubation of the inoculated cells for 16 h perfusion was started with D = 0.36 h-1 corresponding to 0.8·µmax (time 0 h in Fig. (3)). The dilution rate was increased stepwise in intervals of approx. one day up to a dilution rate of D = 1.2 h-1, corresponding to 2.4·µmax. During the first 100 h the cell density in the harvest flow decreased, whilst the pH remained constant at 5.4. At this pH still a significant cell growth can be expected (compare Fig. (2)). Lactose was completely consumed for all dilution rates. Lactate concentration in the harvest flow was constant at 7.9 g·L-1 resulting in constant volume specific lactate production rates for dilution rates of D = 0.42 h-1 and D = 0.72 h-1.
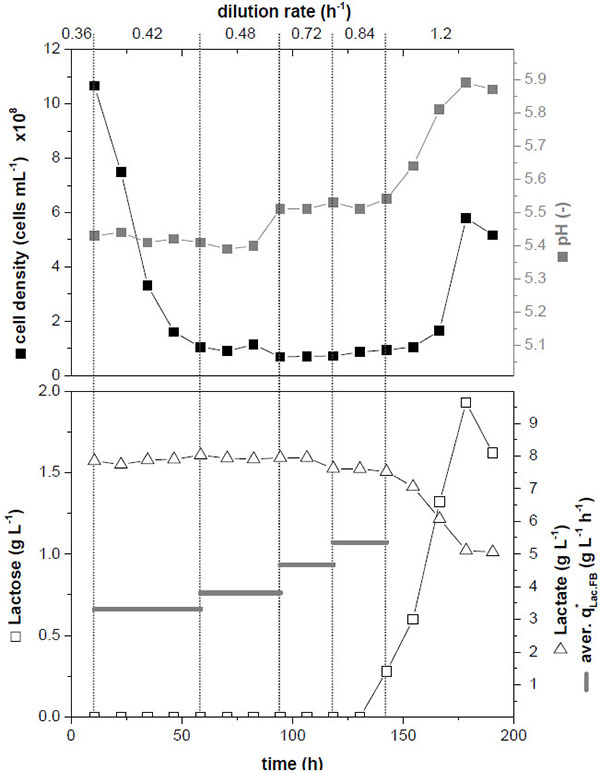
When the dilution rate was further increased to D = 0.84 h-1 and D = 1.2 h-1, cell density, pH and lactose concentration in the harvest flow increased, lactate concentration and volume specific lactate productivity decreased. Obviously a wash-out of cells occurred. This effect was not expected at these low dilution rates, as in earlier fixed-bed cultivations with L. lactis steady-states could be maintained up to D = 4 h-1 [7]. In that case a larger carrier [CERAMTEC, see Introduction] and a different medium formulation was used. The different size of the carriers could be an explanation for the discrepancy. Fixed bed systems tend to become instable, if channels between the carriers become overgrown by cells resulting in diffusion gradients, e.g. for nutrients, metabolites or pH. This effect is even pronounced for carriers with smaller diameters, as the width of the channels between the carriers is smaller in this case and can be blocked by growing cells. Similar effects have been observed for mammalian cells grown in fixed bed reactors [19]. It was decided to use a larger fraction of 8 mm mean diameter of the VitraPOR® carrier for the following experiment in the 1 L-fixed-bed bioreactor for reasons mentioned in the material section.
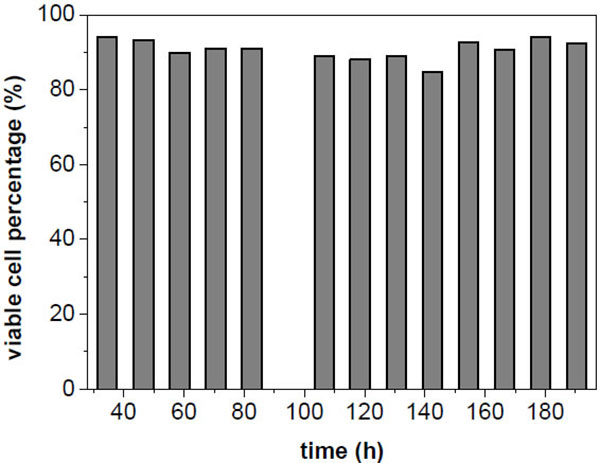
The viability of the cells in the harvest flow remained around 85 - 93% during the whole cultivation (Fig. 4). This can be regarded as high, as similar values were found at the end of exponential growth phase in batch cultures (data not shown).
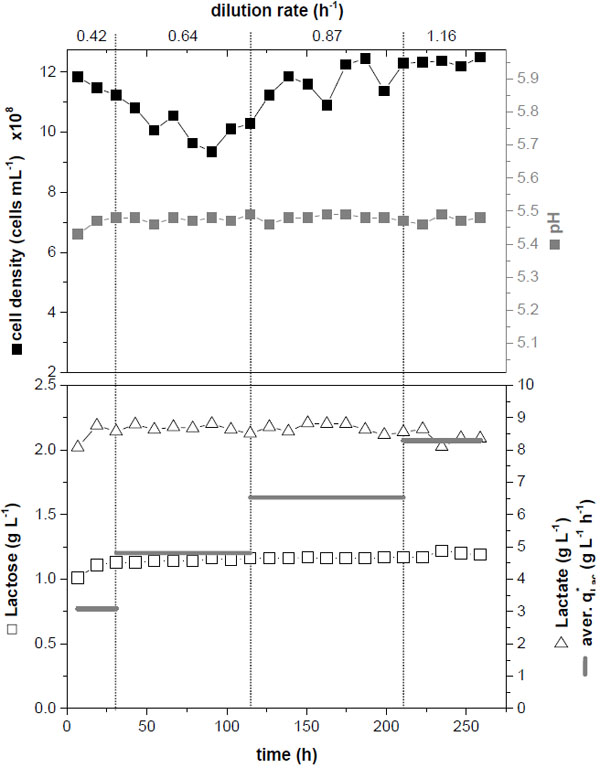
Finally, performance and long-term stability in the 1 L-fixed-bed bioreactor were investigated by increasing the dilution rate step-wise (Fig. 5). After incubation of the inoculated cells for 18 h perfusion was started with D = 0.42 h-1 corresponding to 0.8·µmax (time 0 h in Fig. (5)). The dilution rate was increased stepwise in intervals of approx. 3 – 4 days up to a dilution rate of D = 1.16 h-1, corresponding to 2.4·µmax. The cell density in the harvest flow (approx. 109 cells mL-1) and the pH (approx. 5.8) remained constant. At this pH a significant cell growth can still be expected (compare Fig. (2)). Lactose was not completely consumed, but remained constant at the different dilution rates (approx. 1 g L-1); lactate concentration in the harvest flow was constant (approx. 9 g L-1) as well, resulting in constant volume specific lactate production rates. The highest value (8.3 g L-1 h-1) was determined for D = 1.16 h-1. In contrast to the experiment in the 100 mL-bioreactor (Fig. 3) the cultivation remained stable, probably due to the larger carrier size used here (as discussed above).
Comparison of Suspension and Fixed-bed Systems on Different Scales
For comparison of suspension and fixed-bed cultures the volume specific lactate productivity was plotted against the dilution rate (Fig. 6). For batch culture a value of 1 g L-1 h-1 was obtained. Continuous suspension cultures in chemostat mode showed the expected course [7]. At first the productivity increased with increasing dilution rate up to a maximum of 2.3 g L-1 h-1. When the dilution rate got close to the maximal specific growth rate µmax, the productivity decreased, as wash-out of cells occurred. If dilution rates larger than the maximal specific growth rate are applied in chemostat cultures without cell retention, cells will be definitely to washed out.
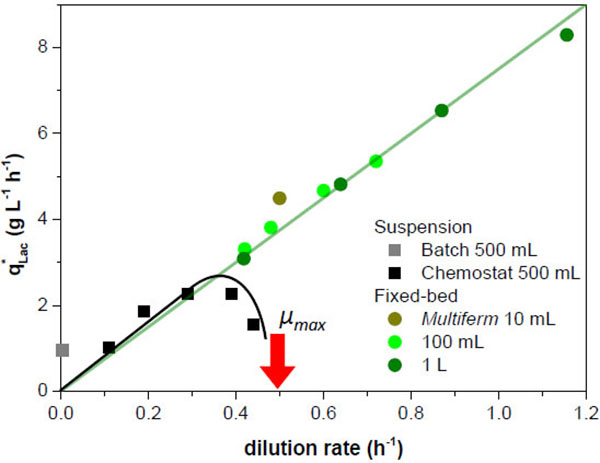
For the fixed-bed-cultures, by definition a perfusion culture with cell retention, the productivity increased steadily with rising dilution rate. At low dilution rates (up to approx. D = 3 h-1) similar productivities were found as in chemostat cultures. But in contrast to the chemostat culture the productivity increased further with increasing dilution rate. Up to the highest dilution rate applied here (D = 1.16 h-1, corresponding to 2.4·µmax) the curve did not flatten. The highest value determined here is approx. three times higher than the maximum in chemostat cultivation. Obviously the maximum for fixed-bed cultures has not been reached so far. This is in accordance with own observations reported in [7]. All fixed-bed systems used here can be described by the same spline.
CONCLUSION
The goal of the study was to evaluate the performance of fixed-bed bioreactor systems on different scales compared to the suspension culture. For this data for different suspension and fixed-bed reactors obtained with one specific strain cultivated in the same medium were used. The suggested concept for the development of fixed-bed processes could be confirmed. The first step, the multi-fixed-bed bioreactor Multiferm, provides an ideal downscaled and economical system that can be used for basic studies with low demands on equipment, medium, cells and labor. The next step, a 100 mL fixed-bed system provides data on the performance and long-term-stability of the culture. Problems that might not have been shown up in the Multiferm, e.g. insufficient long-term stability, can be detected here. The next further step, the 1 L fixed-bed, can be regarded as pilot scale already, as for the highest dilution rate applied here the medium throughput was already at 27.6 L per day. Even higher dilutions rates seem to be possible [7]. It is important to highlight that the data for all three fixed bed scales could be described by the same spline. This is very important with respect to scale-up, as obviously data from small scale systems can be used to predict the performance on a larger scale (for more details on scale-up see [7]).
As expected, fixed-bed bioreactors could be operated in a perfusion mode at a steady-state with dilution rates much higher than the maximum specific growth rate. Obviously the productivity in the fixed bed systems had not yet reached the maximum value. Therefore, a very high volume specific lactate productivity can be reached and maintained for long periods of time [7]. Especially long-term stability is the main premise for acceptance of continuous cultures on industrial scale. Alternative perfusion systems, mainly suspension cultures coupled with cell retention devises (e.g. filtration units) show significant limitations in this respect.
The fixed-bed process with lactic acid bacteria on macroporous carriers could be transferred to pilot scale without loss in productivity. The radial-flow bed geometry offers a further scale-up of the fixed-bed concept. Fixed-bed units with a radius in the range of the 1 L-reactor applied here can be extended in length. Several units can be operated in parallel. In conclusion, a process development tool for fixed-bed processes is now at hand that will pave the way for an industrial application of this promising technology.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
The financial support from Federal Ministry of Education and Research (BMBF) under grant No. 0.31A124A (“ProTool 2”) is gratefully acknowledged just as the work of the project partners (Ingenieurbüro Dr.-Ing. Schoop, medorex e.K. and Prof. Dr.-Ing. Volker C. Hass from Hochschule Furtwangen). The project was part of the BMBF BIOKATALYSE2021 cluster.


