All published articles of this journal are available on ScienceDirect.
Microcalorimetric Investigation of the Effect of the Ionic Liquid 1-Butyl-3-Methylimidazolium Chloride on the Fermentation of Saccharomyces cerevisiae AY93161 for Lignocellulosic Ethanol Production
Abstract
The effects of ionic liquid 1-butyl-3-methylimidazolium chloride (BMIMCl) on the ethanol fermentation process of Saccharomyces cerevisiae AY93161 were investigated by using microcalorimetry. On the basis of microcalorimetric and process data, the thermokinetic parameters of the ethanol fermentation process at different BMIMCl concentrations from 0.001 to 5 gL-1 were calculated. Compared to the control, the BMIMCl caused a decreased value of the maximum specific growth rate µm (from 0.226 to 0.105 h-1), and an increased value of the maximum specific produced heat rate pm (from 2.08 to 7.06 mWlg-1) and the total heat output H for producing 1 g ethanol (from 990 to 1871 Jg-1). The decreased µm and increased pm and H led to lower final yeast concentration (from 3.85 to 2.39 gL-1) and ethanol concentration (from 40.3 to 25.1 gL-1). This gives useful information for improving the lignocellulosic ethanol production process using the ionic liquid technology.
INTRODUCTION
Production of sustainable bio-energy has focused on the research towards renewable resources as an alternative to depletion of fossil resources as well as growing environmental issues such as emission of green house gases and air pollution by incomplete combustion of fossil fuels [1]. Lignocellulosic biomass is the most abundant and renewable natural resource in the world. It has the potential to serve as a renewable feed-stock to chemical commodity and production of fuel, particularly bio-ethanol [2]. Production of ethanol generally consists of two sub-processes: conversion of carbohydrates in lignocellulosic biomass to fermentable sugars and fermentation of the obtained sugars to ethanol. Because of the complex structure of lignin and hemicellulose and cellulose in lignocellulosic biomass, preparation of fermentable sugars is often a challenging work during lignocellulosic ethanol production [2]. Use of ionic liquids has provided a new technical tool to convert the carbohydrates in lignocellulosic biomass to fermentable sugars for ethanol production [3]. A number of studies indicate that the carbohydrates in lignocellulosic biomass can be efficiently converted to fermentable sugars by using the ionic liquid technology [4-6]. However, it is inevitable that some ionic liquids are remained in the obtained sugars. Some previous researches have shown that the residual ionic liquids in sugars have a negative effect on the subsequent ethanol fermentation process, especially for yeast growth [7-9]. The toxicity studies on ionic liquids to yeast cells demonstrate that the interactions between ionic liquids and yeast mitochondrial membrane protein can lead to the morphology change of yeast and its mitochondria, alter the polarization of its mitochondrial membrane potential, and shift its metabolism from respiration to fermentation [10, 11]. Therefore, alleviating the toxicity of residual ionic liquids to yeast growth and their negative effects on the subsequent fermentation process is a key issue for lignocellulosic ethanol production using the ionic liquid technology. To do so, it is essential to understand the metabolic regulation mechanism of the residual ionic liquids affecting ethanol fermentation process and its thermokinetics.
Microcalorimetry is a laboratory method for real-time, continuous measurement of the heat flow rate and cumulative amount of heat consumed or produced during a biological process [12]. As a universal, integral, non-destructive, and highly sensitive method, microcalorimetry is often used to online monitor the bacterial growth and metabolism for a microbial fermentation process [13]. At present, microcalorimetry has been widely used to study thermokinetics for a biological process [12-14]. In this work, the effects of ionic liquid 1-butyl-3-methylimidazolium chloride (BMIMCl) on the ethanol fermentation process of Saccharomyces cerevisiae AY93161 were investigated by using microcalorimetry in combination with the conventional microbial measurements. Based on the microcalorimetric and process data, the metabolic regulation mechanism of the BMIMCl affecting ethanol fermentation process and its thermokinetics were analyzed. This will give useful information for improving the lignocellulosic ethanol production process using the ionic liquid technology by alleviating the negative effect of residual ionic liquids on the subsequent fermentation process.
MATERIALS AND METHODS
All experiments were done in triplicate, and all experimental procedures were performed under aseptic conditions.
Chemicals
The BMIMCl used in this study was obtained from Lanzhou Greenchem ILs, LICP, CAS, China and its purity was 99% up based on HPLC analysis. All other chemicals employed in this study were of reagent grade and purchased from Wuhan Zhenchun Biological Technology Co. Ltd., China.
Inoculum Preparation
The yeast Saccharomyces cerevisiae AY93161 was used throughout this study. The stock cultures were maintained on YPD agar plates at 4 °C and transferred to fresh plates every 4 weeks to avoid micro-organism degradation. The inoculum preparation was by means of micro-organism transfer from stock cultures to a fresh plate and grew for 48 h at 30°C. Following this period, single colonies were transferred to a 250 ml flask with 100 ml YPD medium. The flask was placed on an orbital shaker with a shaking diameter 5 cm and a shaking frequency 200 rpm and incubated at 30°C for 24 h. This was used as the inoculum for microcalorimetric measurements and parallel ethanol fermentation experiments, and its yeast concentration is about 1.5× 1011 cells l-1.
Media Compositions
The composition of the YPD agar medium was as follows: D-glucose 20 gL-1, peptone 20 gL-1, yeast extract 10 gL-1, and agar 15 gL-1.
The composition of the YPD medium was as follows: D-glucose 20 gL-1, peptone 20 gL-1, and yeast extract 10 gL-1.
The composition of the ethanol fermentation medium was as follows: D-glucose monohydrate 100 gL-1, peptone 20 gL-1, and yeast extract 10 gL-1.
Each medium was autoclaved at 121°C for 20 minutes after the suitable amount of BMIMCl was added to a given concentration.
Ethanol Fermentation
In a 250 ml glass bottle, 96 ml ethanol fermentation medium with a given BMIMCl concentration and 4 ml inoculum were added. Then the suspension in the bottle was intensively stirred. After that, 5 ml fermentation broth from the bottle was put into a 20 ml calorimetric ampule, the ampule was sealed and used for later microcalorimetric measurement. The bottle with the remaining 95 ml suspension was also sealed and used for the parallel ethanol fermentation. As the parallel experiment of microcalorimetric measurement, the bottle was placed into a thermostat at 30°C and kept ethanol fermentation under the same conditions as microcalorimetric measurements. During the fermentation process, small samples were taken at regular intervals for later analytical usage.
Microcalorimetric Measurement
A TAM Air isothermal microcalorimeter (Thermometric AB, Sweden), which was a multichannel microcalorimetric system, was used to record the heat flow rate of the ethanol fermentation process, which was calibrated by the release of electrical energy in a resistor and the heat effect of the sample ampoule. The heat power detection limit is stated to be ±2μW. The detailed structure and operation of the instrument have been described previously [14]. All microcalorimetric experiments were performed at 30°C using the ampule method. The 20 ml sealed ampules with 5 ml ethanol fermentation broth, which were prepared as described above, were used for these microcalorimetric measurements. The power time curves of the ethanol fermentation process were recorded by a computer.
Analytical Methods
The samples taken from the ethanol fermentation process were used to determine the concentration of yeast, ethanol and the fermentable sugars. Yeast concentration was determined by the dry weight method [7]. Ethanol content was determined by gas chromatography [15] and the fermentable sugars concentration was estimated using the 3,5-dinitrosalicylic acid method [16].
RESULTS AND DISCUSSION
Effect of BMIMCl on the Ethanol Fermentation Process
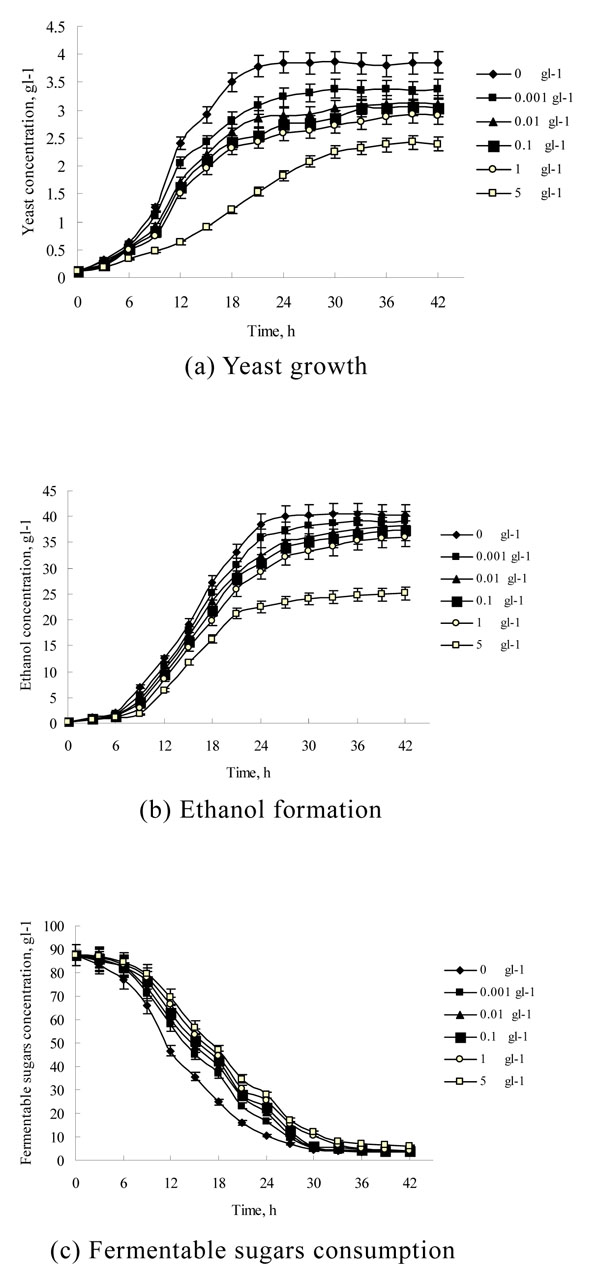
The BMIMCl inhibits yeast growth and has a negative effect on ethanol production [7, 8]. Under different fermentation conditions, the BMIMCl has a different influencing degree on the ethanol fermentation process [7, 8]. In order to have a correct understanding on microcalorimetric data, the ethanol fermentation was carried out under the same conditions as the microcalorimetric measurement in this work. The time courses of yeast growth, ethanol production and the fermentable sugars consumption at different BMIMCl concentrations for ethanol fermentation process are shown in Fig. (1). As shown in Fig. (1), the ethanol fermentation had the characteristics of the typical batch fermentation process at all BMIMCl concentrations. The yeast growth during ethanol fermentation process consisted of four different periods: the lag phase, the log growth phase, the late log growth phase, and the stationary phase. Compared with the control, the lag and log growth phase cost almost the same time at all BMIMCl concentrations, but the time of the late log growth phase and the integral fermentation time were extended with increase of BMIMCl concentration. When the BMIMCl concentration was 0, 0.001, 0.01 and 1 gL-1, the yeast reached its highest concentration at 21, 27, 30, 33 and 36 h, and the produced ethanol arrived at its highest concentration at 27, 30, 33, 36 and 39 h respectively. When the BMIMCl concentration was 5 gL-1, the yeast and ethanol concentration was slowly increased at 42 h. Table 1 lists some important ethanol fermentation process data at different BMIMCl concentrations. As indicated in Table 1, when the BMIMCl concentration increased from 0 to 1 gL-1, the final yeast and ethanol concentration, the ethanol yield from the fermentable sugars (Yp/s) and the maximum yeast specific growth rate (µm) all decreased, but the final fermentable sugars concentration and the average specific ethanol productivity of yeast (E) was slightly increased. All these results suggest that the negative effect of BMIMCl on ethanol production came from its inhibition on yeast growth. This is consistent with our previous studies [7, 8]. From Fig. (1) and Table 1, it is also found that the BMIMCl severely inhibited yeast growth and ethanol formation when its concentration reached 5 gL-1. Its final yeast and ethanol concentration was much lower than that when the BMIMCl concentration was 1 gL-1. It implies that high BMIMC1 concentrations may cause a change in the yeast metabolism. This is consistent with the previous studies that ionic liquids could shift yeast metabolism from respiration to fermentation and damage mitochondrial function by inducing hyperpolarization of the mitochondrial membrane [10, 11].
Thermokinetic Analysis on the Ethanol Fermentation Process at Different BMIMCl Concentrations
Ethanol fermentation is a biological process which converts the fermentable sugars to ethanol and, at the same time, produces large amounts of heat. Microcalorimetry can easily record the thermogenic power-time curves of the ethanol
| Ci (gL-1) | 0 | 0.001 | 0.01 | 0.1 | 1 | 5 |
|---|---|---|---|---|---|---|
| Cb (gL-1) | 3.85±0.12 | 3.38±0.08 | 3.11±0.10 | 3.04±0.06 | 2.90±0.09 | 2.39±0.05 |
| Cs (gL-1) | 3.4±0.2 | 3.6±0.3 | 3.7±0.4 | 3.9±0.4 | 3.8±0.3 | 6.1±0.5 |
| Cp (gL-1) | 40.3±0.7 | 39.1±0.6 | 38.2±0.8 | 37.3±0.3 | 35.9±0.7 | 25.1±0.5 |
| Yp/s | 0.460±0.008 | 0.446±0.007 | 0.436±0.009 | 0.426±0.003 | 0.410±0.008 | 0.287±0.006 |
| E | 10.47±0.19 | 11.57±0.18 | 12.28±0.27 | 12.27±0.10 | 12.37±0.24 | 10.50±0.21 |
| µm (h-1) | 0.226±0.012 | 0.216±0.008 | 0.193±0.006 | 0.190±0.005 | 0.186±0.007 | 0.105±0.006 |
| Tm (min) | 766±16 | 773±12 | 765±18 | 766±9 | 763±14 | 762±17 |
| Pm (mW) | 5.02±0.12 | 4.32±0.08 | 3.94±0.05 | 3.91±0.07 | 4.37±0.03 | 4.38±0.06 |
| Cbm (gL-1) | 2.41±0.08 | 2.05±0.07 | 1.72±0.03 | 1.61±0.06 | 1.50±0.06 | 0.62±0.04 |
| Cpm (gL-1) | 12.5±0.5 | 11.1±0.3 | 10.3±0.6 | 9.4±0.4 | 8.6±0.7 | 6.4±0.2 |
| pm (mWlg-1) | 2.08±0.05 | 2.11±0.04 | 2.29±0.03 | 2.43±0.04 | 2.91±0.02 | 7.06±0.10 |
| K (h-1) | 0.233±0.015 | 0.207±0.009 | 0.180±0.010 | 0.185±0.006 | 0.196±0.008 | 0.201±0.012 |
| Qtotal (J) | 199.5±4.8 | 200.4±3.6 | 208.9±2.7 | 228.0±4.3 | 235.4±5.6 | 234.8±7.2 |
| Qlog (J) | 72.1±1.5 | 65.1±0.9 | 67.4±1.2 | 68.2±0.8 | 72.4±2.3 | 69.0±1.6 |
| HY (Jlg-1) | 8.15±0.09 | 10.03±0.91 | 14.99±0.84 | 20.78±2.92 | 26.87±2.57 | 54.71±0.37 |
| HE (Jlg-1) | 4.18±0.16 | 4.18±0.16 | 4.18±0.16 | 4.18±0.16 | 4.18±0.16 | 4.18±0.16 |
| H (Jg-1) | 990±24 | 1025±18 | 1094±14 | 1223±23 | 1311±31 | 1871±57 |
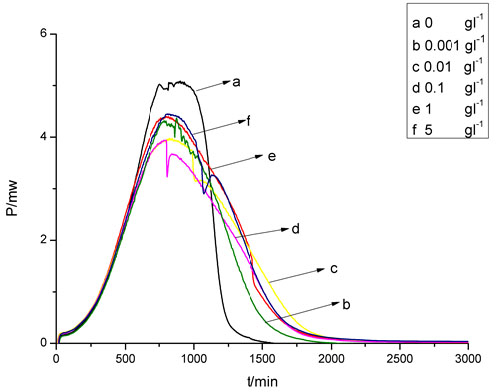
fermentation process. The power-time curves can provide considerable kinetic and thermodynamic information about the ethanol fermentation process that is unavailable in other methods. The power-time curves of the ethanol fermentation process at different BMIMCl concentrations are shown in Fig. (2). From these power-time curves, it is found that the heat output of ethanol fermentation process at all BMIMCl concentrations included four different periods: the slow heat producing phase, the log heat producing phase, the stationary heat producing phase, and the declining heat producing phase. The slow and log heat producing heat phase was in good agreement with the lag and log growth phase during the ethanol fermentation process in the parallel experiments, and the stationary and declining heat producing phase was corresponding to the late log growth and the stationary phase. As shown in Fig. (2), for all BMIMCl concentrations, the heat producing power reached the maximum almost at the same time, but the integral fermentation time was extended with the BMIMCl concentration increasing. Based on the microcalorimetric measurement, some important thermokinetic parameters of ethanol fermentation process at different BMIMCl concentrations could be obtained and were listed in Table 1. As indicated in Table 1, the time of the maximum producing heat power (Tm) at all BMIMCl concentrations was almost simultaneous, and at Tm, the yeast and ethanol concentration decreased, but the maximum specific producing heat power (pm) increased with the BMIMCl concentration increasing, which led to the maximum producing heat power (Pm) decreased at the BMIMCl concentration from 0 to 0.1 gL-1, but it increased at the BMIMCl concentration from 0.1 to 5 gL-1. The producing heat power constant (K) at a given BMIMCl concentration was calculated as described by Braissant et al. [12], but it was not equal to the µm, which indicated the specific producing heat power changed during the ethanol fermentation process. The heat output for log phase (Qlog) and the total heat output for the integral ethanol fermentation process (Qtotal) could be obtained from the power-time curves as described by Yao et al. [14]. Based on the Qlog and Qtotal, the heat output for yeast concentration increasing 1 gL-1 (Hy) and the heat output for ethanol concentration increasing 1 gL-1 (HE) could be easily calculated. The HE was the same for all BMIMCl concentrations, but the Hy increased with the BMIMCl concentration increasing, which caused the increase in the total heat output for producing 1 g ethanol (H) when the BMIMCl concentration increases. It is noteworthy that Hy and H were much higher at BMIMCl concentration 5 gL-1 than 1 gL-1. This gave another evidence that high BMIMCl concentration might cause a change in the yeast metabolism. Some previous studies have indicated the BMIMCl inhibited yeast growth because of its interaction with the yeast mitochondrial membrane protein [7, 8, 10, 11]. This interaction could lead to more heat output for yeast growth and maintenance, which might be the reason why H increases with the BMIMCl concentration increasing. Base on the above analysis, the possible influencing mechanism of the BMIMCl on ethanol fermentation process was the interaction between the BMIMCl and the yeast mitochondrial membrane protein inhibited yeast growth [10, 11, 17, 18], more fermentable sugars were consumed for yeast growth and maintenance, which led to lower final yeast and ethanol concentration, lower ethanol yield from the fermentable sugars and longer fermentation time. It also caused that the H increases with the BMIMCl concentration increasing. These results indicate that residual concentrations of ionic liquids affect the fermenting yeast cell metabolism and its thermokinetics, hence strategies to reduce their effects on the biocatalyst are required for optimizing product yields using the ionic liquid technology.
CONCLUSION
The negative effects of BMIMCl on the ethanol fermentation process of Saccharomyces cerevisiae AY93161 were investigated via microcalorimetry, and its metabolic regulation mechanism was analyzed based on its thermokinetic data. The BMIMCl decreased µm and increased pm and H, which caused lower final yeast and ethanol concentration. This provides useful information for the improvement of the lignocellulosic ethanol production process by using the ionic liquid technology.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
This work was supported by the National Natural science Foundation of China No.21176196, Graduate Innovative Fund of Wuhan Institute of Technology CX2015077, Key Laboratory for Green Chemical Process of Ministry of Education GCP201501 and Hubei Key Laboratory of Novel Chemical Reactor and Green Chemical Technology NRGCT201501.


